QUESTION IMAGE
Question
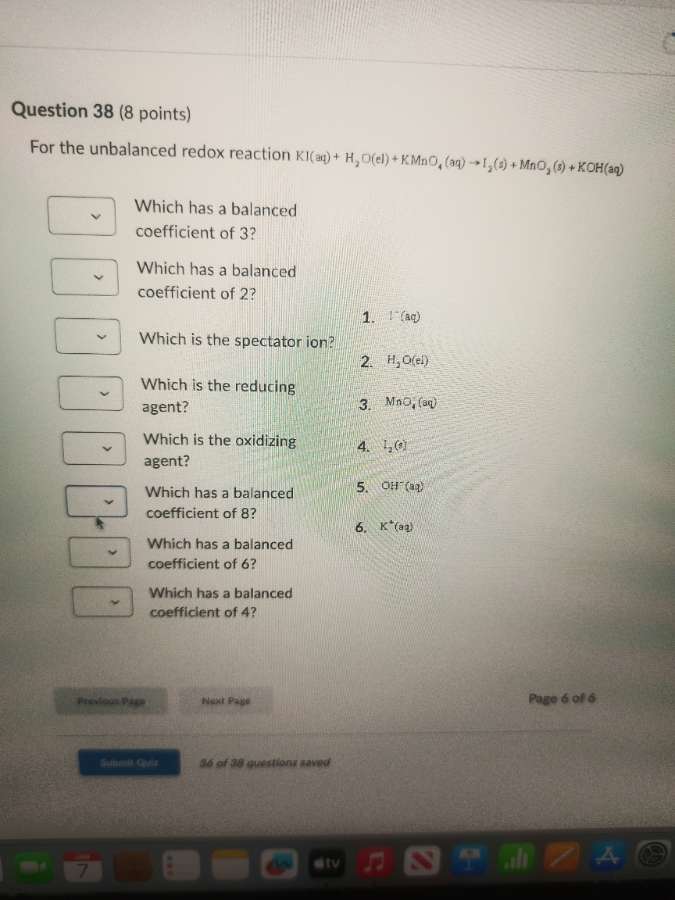
question 38 (8 points)
for the unbalanced redox reaction ki(aq) + h₂o(l) + kmno₄(aq) → i₂(s) + mno₂(s) + koh(aq)
which has a balanced coefficient of 3?
which has a balanced coefficient of 2?
which is the spectator ion?
which is the reducing agent?
which is the oxidizing agent?
which has a balanced coefficient of 8?
which has a balanced coefficient of 6?
which has a balanced coefficient of 4?
- i⁻(aq)
- h₂o(l)
- mno₄⁻(aq)
- i₂(s)
- oh⁻(aq)
- k⁺(aq)
To solve this, we first balance the redox reaction. Let's identify oxidation and reduction half - reactions:
Step 1: Identify Oxidation and Reduction
- Oxidation (I⁻ to I₂): The oxidation state of I in KI (I⁻) is - 1, and in I₂ it is 0. So, 2I⁻→I₂ + 2e⁻ (loss of electrons, oxidation).
- Reduction (MnO₄⁻ to MnO₂): The oxidation state of Mn in MnO₄⁻ is + 7, and in MnO₂ it is + 4. So, MnO₄⁻+ 2H₂O+ 3e⁻→MnO₂ + 4OH⁻ (gain of electrons, reduction).
Step 2: Balance Electrons
To balance the electrons, we multiply the oxidation half - reaction by 3 and the reduction half - reaction by 2.
- Oxidation: \(6I^-(aq)
ightarrow3I_2(s)+6e^-\)
- Reduction: \(2MnO_4^-(aq)+4H_2O(l)+6e^-
ightarrow2MnO_2(s)+8OH^-(aq)\)
Step 3: Combine Half - Reactions and Balance the Whole Reaction
Adding the two half - reactions together:
\(6KI(aq)+2KMnO_4(aq)+4H_2O(l)
ightarrow3I_2(s)+2MnO_2(s)+8KOH(aq)\)
Now let's answer each part:
1. Which has a balanced coefficient of 3?
From the balanced reaction, the coefficient of \(I_2(s)\) (option 4) is 3.
2. Which has a balanced coefficient of 2?
From the balanced reaction, the coefficient of \(MnO_4^-(aq)\) (option 3) and \(MnO_2(s)\) is 2. Also, the coefficient of \(KMnO_4\) (which provides \(MnO_4^-\)) is 2.
3. Which is the spectator ion?
In the reaction, \(K^+(aq)\) (option 6) is a spectator ion because it does not participate in the redox process (its oxidation state remains + 1 throughout the reaction).
4. Which is the reducing agent?
The reducing agent is the species that gets oxidized. \(I^-(aq)\) (option 1) is oxidized (loses electrons), so it is the reducing agent.
5. Which is the oxidizing agent?
The oxidizing agent is the species that gets reduced. \(MnO_4^-(aq)\) (option 3) is reduced (gains electrons), so it is the oxidizing agent.
6. Which has a balanced coefficient of 8?
From the balanced reaction, the coefficient of \(OH^-(aq)\) (option 5) is 8.
7. Which has a balanced coefficient of 6?
From the balanced reaction, the coefficient of \(KI\) (which provides \(I^-\)) and the number of electrons transferred (but in terms of species, the coefficient of \(I^-\) from \(KI\) is 6) and the coefficient of \(KOH\) is 8, the coefficient of \(H_2O\) is 4, the coefficient of \(KI\) is 6. The coefficient of \(KI\) (which provides \(I^-\), option 1) is 6.
8. Which has a balanced coefficient of 4?
From the balanced reaction, the coefficient of \(H_2O(l)\) (option 2) is 4.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
- Coefficient of 3: 4. \(I_2(s)\)
- Coefficient of 2: 3. \(MnO_4^-(aq)\)
- Spectator ion: 6. \(K^+(aq)\)
- Reducing agent: 1. \(I^-(aq)\)
- Oxidizing agent: 3. \(MnO_4^-(aq)\)
- Coefficient of 8: 5. \(OH^-(aq)\)
- Coefficient of 6: 1. \(I^-(aq)\) (from \(KI\))
- Coefficient of 4: 2. \(H_2O(l)\)