QUESTION IMAGE
Question
question 42 of 49
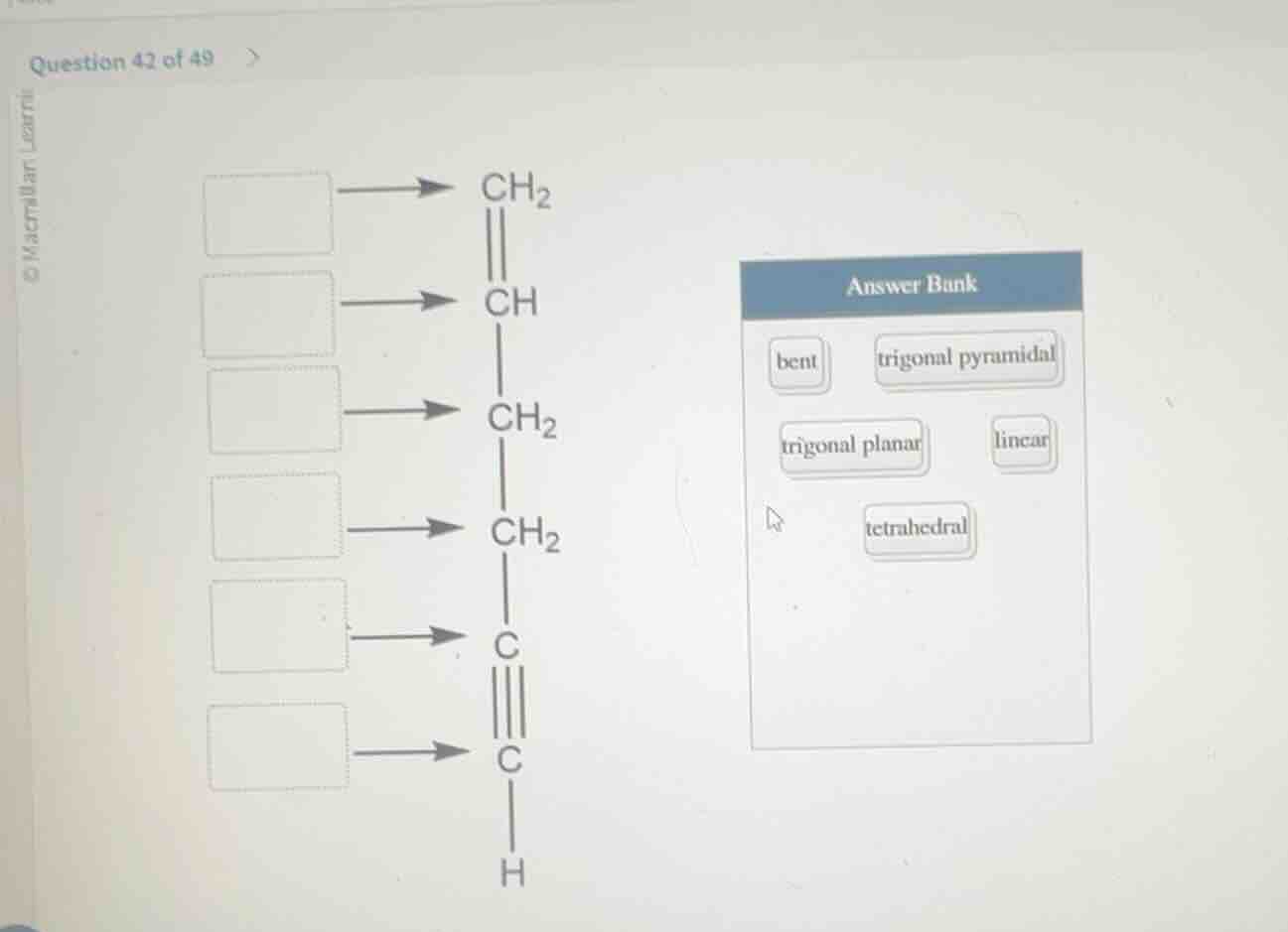
match the molecular geometry (from the answer bank) to each labeled atom in the structure.
answer bank
bent trigonal pyramidal
trigonal planar linear
tetrahedral
structure:
- first ch₂ (double bond c)
- ch (single bond to ch₂, double bond to ch₂? wait, no, the structure is:
ch₂ (double bond to ch)
|
ch (single bond to ch₂)
|
ch₂ (single bond to ch₂)
|
ch₂ (single bond to c with triple bond)
|
c (triple bond to c)
|
c (single bond to h)
wait, the image shows:
top: ch₂ (double bond to ch)
then ch (single bond to ch₂)
then ch₂ (single bond to ch₂)
then ch₂ (single bond to c with triple bond)
then c (triple bond to c)
then c (single bond to h)
arrows point to each of these atoms: ch₂ (double bond), ch, ch₂, ch₂, c (triple bond c), c (single bond h).
answer bank options: bent, trigonal pyramidal, trigonal planar, linear, tetrahedral.
Step1: Analyze top $\text{CH}_2$
The top $\text{CH}_2$ has a carbon with 3 electron domains (1 double bond, 2 single bonds to H), no lone pairs. Electron domain geometry = molecular geometry: trigonal planar.
Step2: Analyze middle $\text{CH}$
The $\text{CH}$ has a carbon with 3 electron domains (1 double bond, 1 single bond to $\text{CH}_2$, 1 single bond to H), no lone pairs. Geometry: trigonal planar.
Step3: Analyze first $\text{CH}_2$ (below $\text{CH}$)
This $\text{CH}_2$ has a carbon with 4 electron domains (4 single bonds: 2 to H, 1 to $\text{CH}$, 1 to $\text{CH}_2$), no lone pairs. Geometry: tetrahedral.
Step4: Analyze second $\text{CH}_2$ (below first $\text{CH}_2$)
This $\text{CH}_2$ has a carbon with 4 electron domains (4 single bonds: 2 to H, 1 to upper $\text{CH}_2$, 1 to $\text{C}$), no lone pairs. Geometry: tetrahedral.
Step5: Analyze upper $\text{C}$ (in triple bond)
This carbon has 2 electron domains (1 triple bond, 1 single bond to $\text{CH}_2$), no lone pairs. Geometry: linear.
Step6: Analyze lower $\text{C}$ (in triple bond)
This carbon has 2 electron domains (1 triple bond, 1 single bond to H), no lone pairs. Geometry: linear.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Top $\text{CH}_2$: trigonal planar
- Middle $\text{CH}$: trigonal planar
- First $\text{CH}_2$ (below $\text{CH}$): tetrahedral
- Second $\text{CH}_2$ (below first $\text{CH}_2$): tetrahedral
- Upper $\text{C}$ (triple bond): linear
- Lower $\text{C}$ (triple bond): linear