QUESTION IMAGE
Question
question 43 of 49
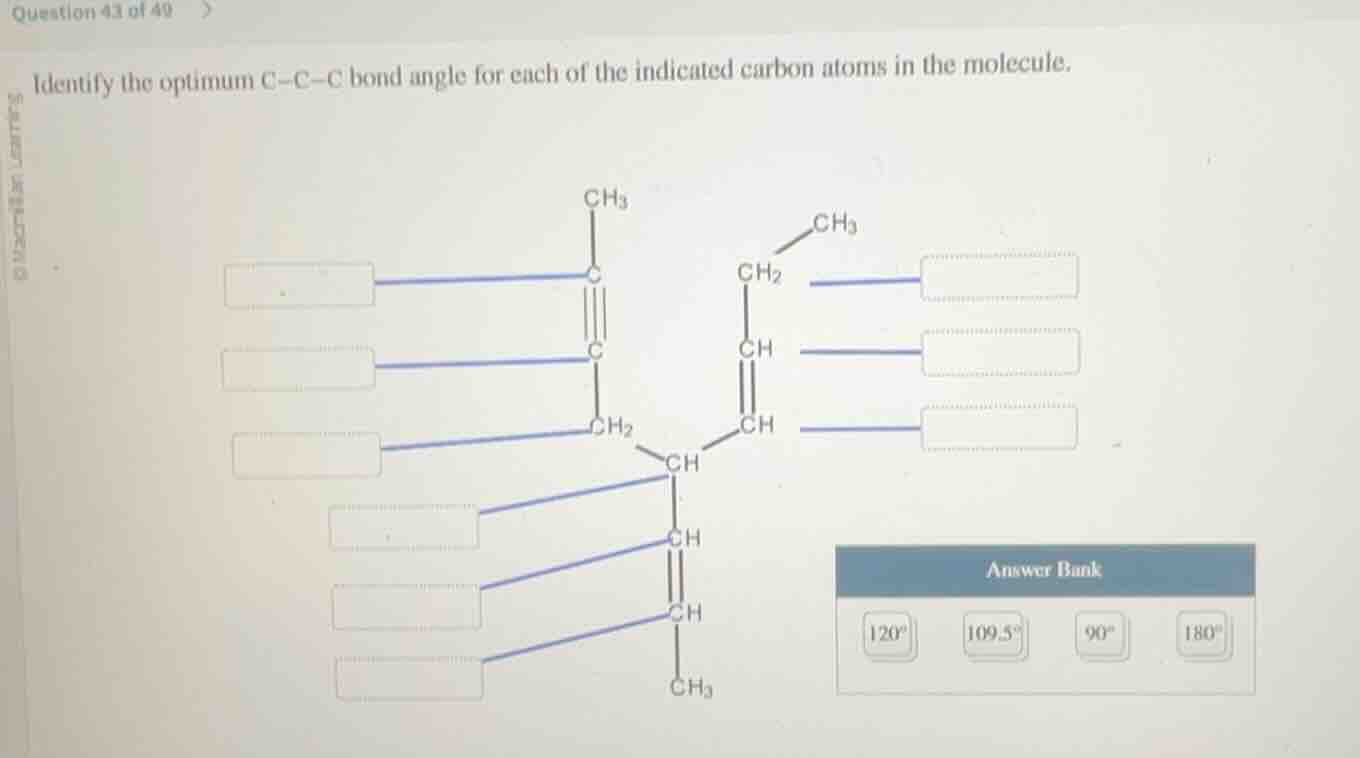
identify the optimum c-c-c bond angle for each of the indicated carbon atoms in the molecule.
(the image shows a molecular structure with carbon atoms and an answer bank with options 120°, 109.5°, 90°, 180°)
Step1: Assign hybridization to each C
Use VSEPR:
- $sp$ (2 electron domains): bond angle $180^\circ$
- $sp^2$ (3 electron domains): bond angle $120^\circ$
- $sp^3$ (4 electron domains): bond angle $109.5^\circ$
Step2: Analyze top alkyne carbons
Leftmost $C$ ($\text{CH}_3\text{C}\equiv$): $sp$ hybridized.
$\text{C-C-C}$ angle: $180^\circ$
Middle alkyne $C$ ($\equiv\text{C-CH}_2-$): $sp$ hybridized.
$\text{C-C-C}$ angle: $180^\circ$
Step3: Analyze $\text{CH}_2$ adjacent to alkyne
$\text{-C-CH}_2-$: $sp^3$ hybridized.
$\text{C-C-C}$ angle: $109.5^\circ$
Step4: Analyze central branched $C$
Central $C$ (connected to 4 groups): $sp^3$ hybridized.
$\text{C-C-C}$ angle: $109.5^\circ$
Step5: Analyze alkene carbons
Right alkene $C$ ($\text{-CH=CH-CH}_2-$): $sp^2$ hybridized.
$\text{C-C-C}$ angle: $120^\circ$
Left alkene $C$ ($\text{-C-CH=CH}-$): $sp^2$ hybridized.
$\text{C-C-C}$ angle: $120^\circ$
Step6: Analyze bottom alkene carbons
Bottom right alkene $C$ ($\text{-C=CH-CH}_3$): $sp^2$ hybridized.
$\text{C-C-C}$ angle: $120^\circ$
Bottom left alkene $C$ ($\text{-CH-C=CH}-$): $sp^2$ hybridized.
$\text{C-C-C}$ angle: $120^\circ$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Topmost (alkyne $\text{CH}_3\text{C}\equiv$): $180^\circ$
- Second top (middle alkyne $\equiv\text{C}-$): $180^\circ$
- Third top ($\equiv\text{C-CH}_2-$): $109.5^\circ$
- Central branched $C$: $109.5^\circ$
- Bottom right alkene ($\text{-CH=CH-CH}_2-$): $120^\circ$
- Bottom middle alkene ($\text{-C-CH=CH}-$): $120^\circ$
- Bottom left alkene ($\text{-CH-C=CH}-$): $120^\circ$
- Bottommost ($\text{-C=CH-CH}_3$): $120^\circ$