QUESTION IMAGE
Question
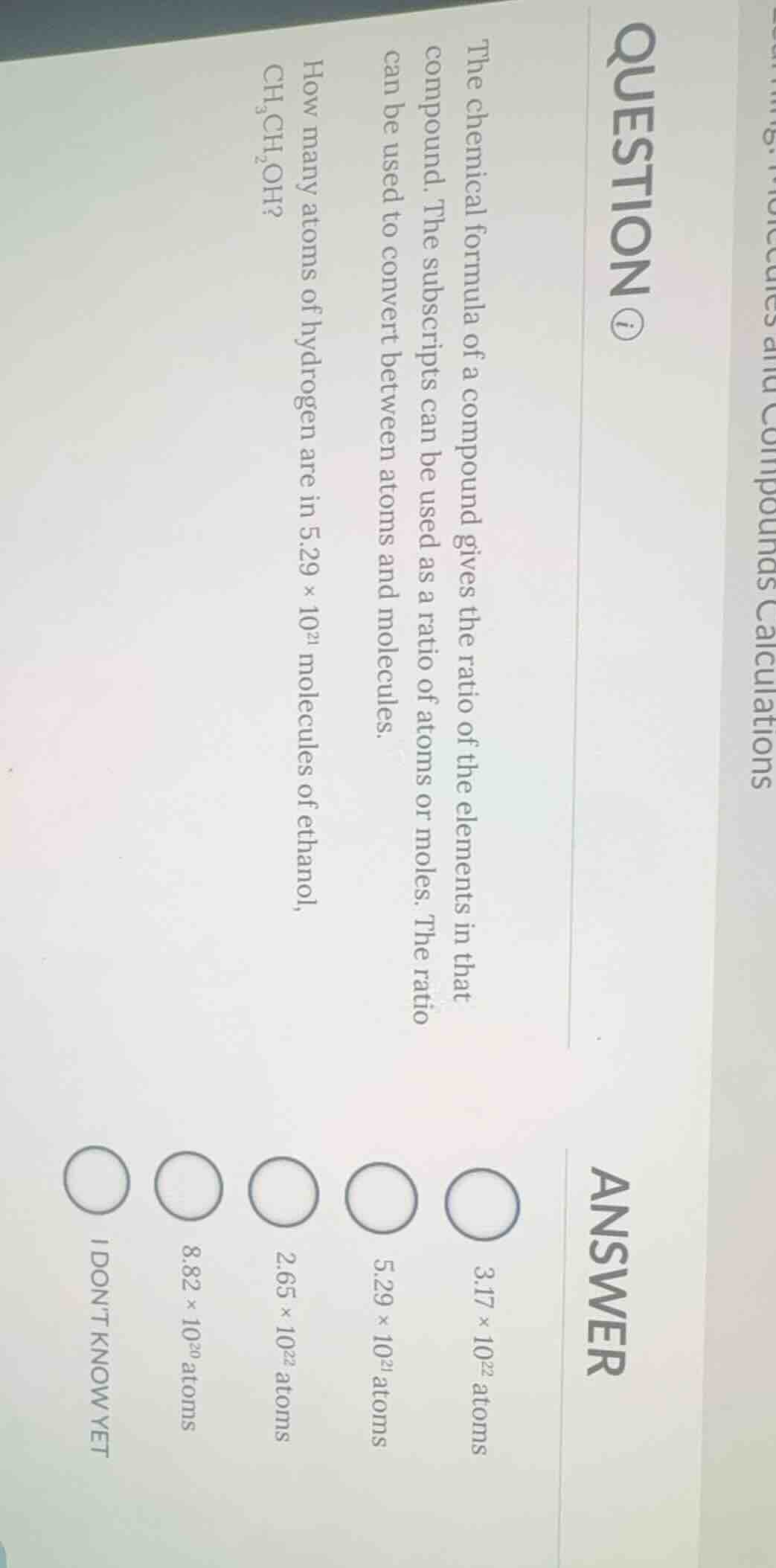
question ⓘthe chemical formula of a compound gives the ratio of the elements in that compound. the subscripts can be used as a ratio of atoms or moles. the ratio can be used to convert between atoms and molecules.how many atoms of hydrogen are in $5.29 \times 10^{21}$ molecules of ethanol, $\text{ch}_3\text{ch}_2\text{oh}$?answer$3.17 \times 10^{22}$ atoms$5.29 \times 10^{21}$ atoms$2.65 \times 10^{22}$ atoms$8.82 \times 10^{20}$ atomsi dont know yet
Step1: Count H atoms per ethanol molecule
From $\text{CH}_3\text{CH}_2\text{OH}$, each molecule has 6 H atoms.
Step2: Calculate total H atoms
Multiply molecule count by H per molecule:
$5.29 \times 10^{21} \times 6$
$= 31.74 \times 10^{21}$
$= 3.174 \times 10^{22} \approx 3.17 \times 10^{22}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3.17 × 10²² atoms