QUESTION IMAGE
Question
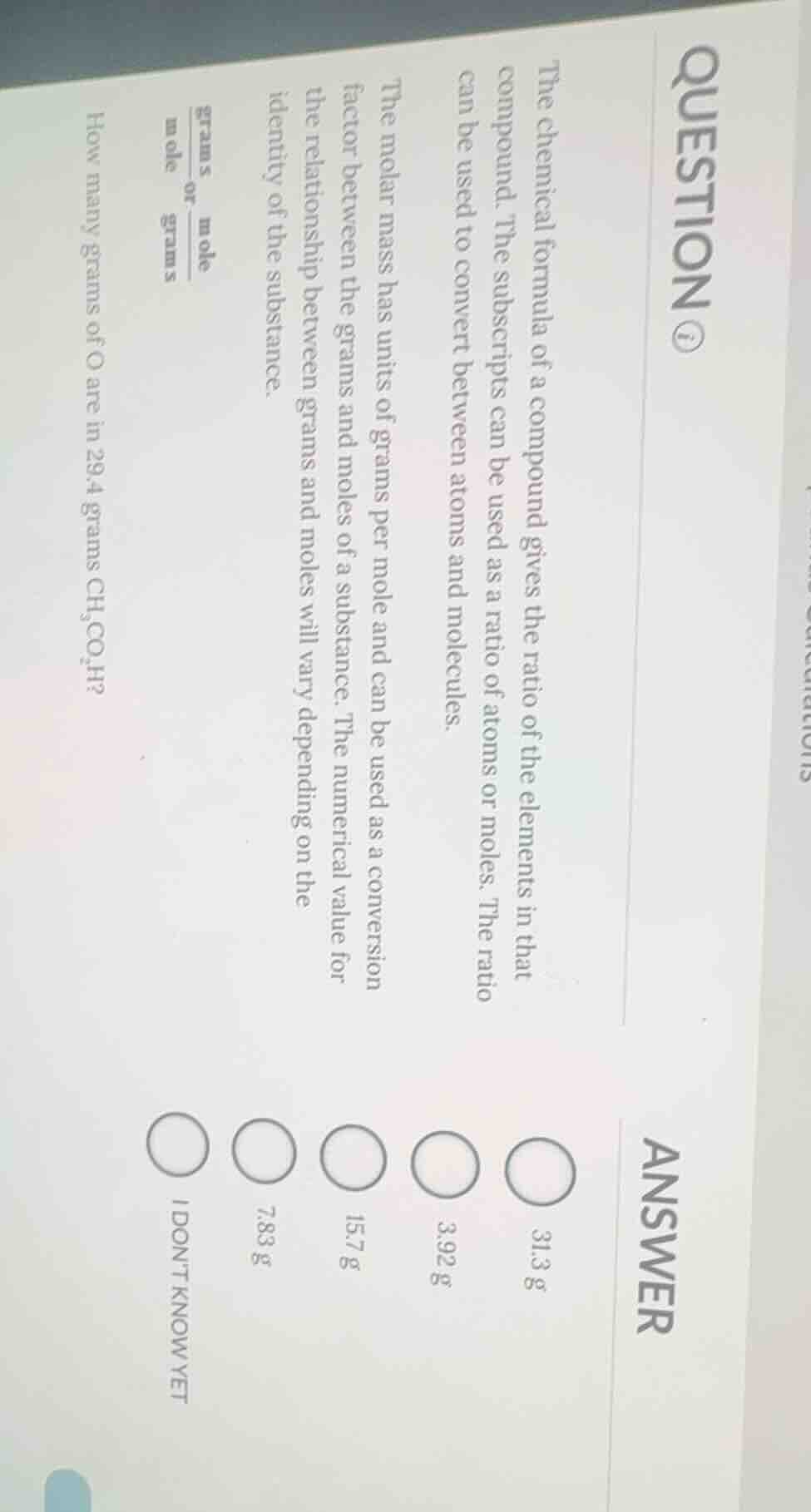
question ①the chemical formula of a compound gives the ratio of the elements in that compound. the subscripts can be used as a ratio of atoms or moles. the ratio can be used to convert between atoms and molecules.the molar mass has units of grams per mole and can be used as a conversion factor between the grams and moles of a substance. the numerical value for the relationship between grams and moles will vary depending on the identity of the substance.$\frac{\text{grams}}{\text{mole}}$ or $\frac{\text{mole}}{\text{grams}}$how many grams of o are in 29.4 grams $\text{ch}_3\text{co}_2\text{h}$?answer$\bigcirc$ 31.3 g$\bigcirc$ 3.92 g$\bigcirc$ 15.7 g$\bigcirc$ 7.83 g$\bigcirc$ i dont know yet
Step1: Calculate molar mass of $\text{CH}_3\text{CO}_2\text{H}$
Molar mass = $2\times12.01 + 4\times1.008 + 2\times16.00 = 60.052\ \text{g/mol}$
Step2: Find moles of $\text{CH}_3\text{CO}_2\text{H}$
$\text{Moles} = \frac{29.4\ \text{g}}{60.052\ \text{g/mol}} \approx 0.4896\ \text{mol}$
Step3: Relate moles of O to compound
1 mole $\text{CH}_3\text{CO}_2\text{H}$ has 2 moles O.
$\text{Moles of O} = 0.4896\ \text{mol} \times 2 = 0.9792\ \text{mol}$
Step4: Calculate mass of O
$\text{Mass} = 0.9792\ \text{mol} \times 16.00\ \text{g/mol} \approx 15.7\ \text{g}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
15.7 g