QUESTION IMAGE
Question
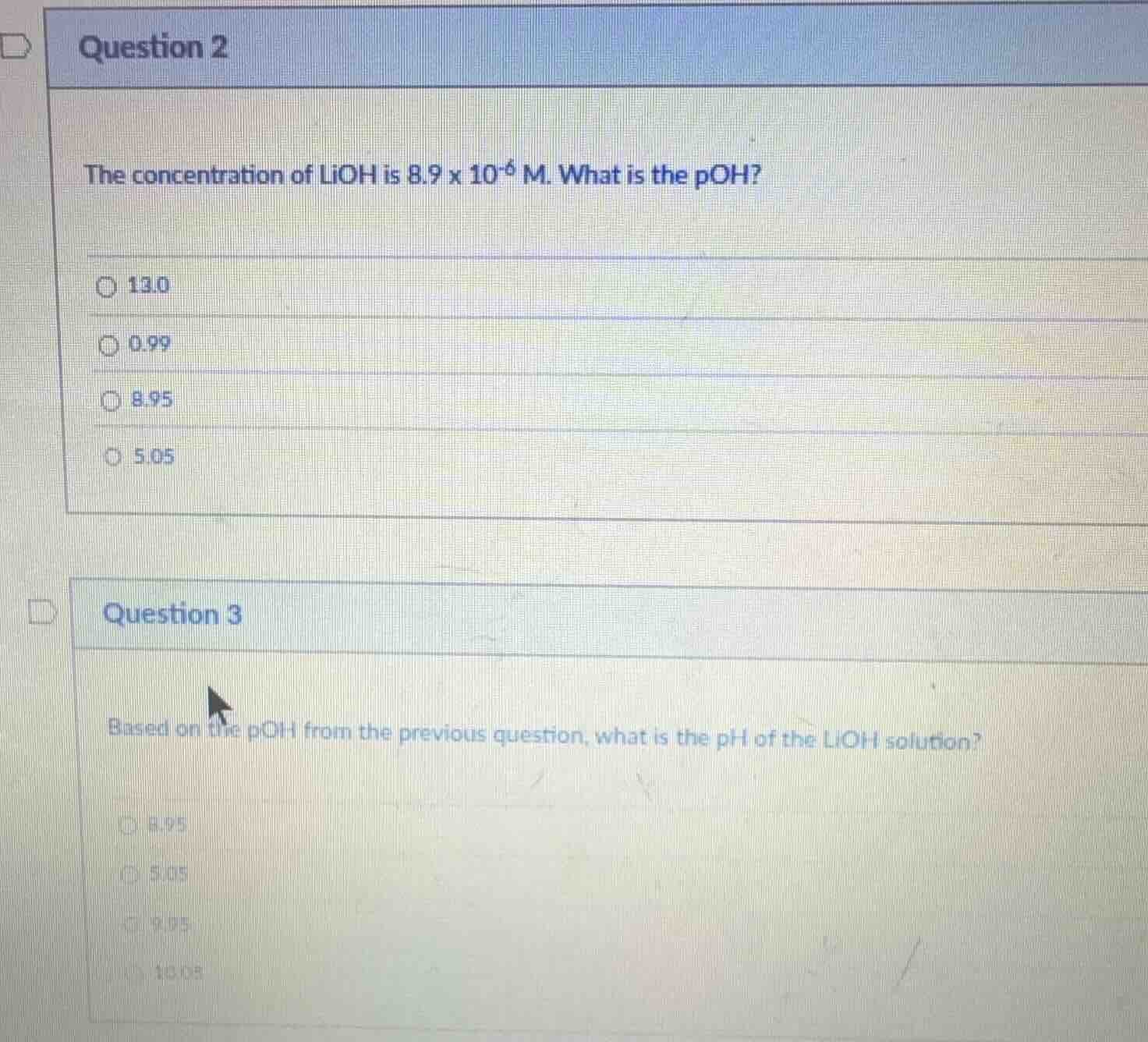
question 2
the concentration of lioh is 8.9 x 10⁻⁶ m. what is the poh?
○ 13.0
○ 0.99
○ 8.95
○ 5.05
question 3
based on the poh from the previous question, what is the ph of the lioh solution?
○ 8.95
○ 5.05
○ 9.05
○ 10.05
Question 2
Step1: Identify $[\text{OH}^-]$ from LiOH
LiOH is a strong base, so it dissociates completely: $\text{LiOH}
ightarrow \text{Li}^+ + \text{OH}^-$. Thus, $[\text{OH}^-] = [\text{LiOH}] = 8.9 \times 10^{-6}\ \text{M}$.
Step2: Calculate pOH using formula
The formula for pOH is $\text{pOH} = -\log_{10}([\text{OH}^-])$. Substitute $[\text{OH}^-] = 8.9 \times 10^{-6}$:
$\text{pOH} = -\log(8.9 \times 10^{-6})$.
Using a calculator, $\log(8.9 \times 10^{-6}) \approx -5.05$, so $-\log(8.9 \times 10^{-6}) \approx 5.05$.
Step1: Recall the relationship between pH and pOH
The relationship is $\text{pH} + \text{pOH} = 14$.
Step2: Substitute pOH from Question 2
From Question 2, pOH = 5.05. So, $\text{pH} = 14 - \text{pOH} = 14 - 5.05 = 8.95$. Wait, no—wait, LiOH is a base, so pH should be greater than 7. Wait, correction: Wait, 14 - 5.05 is 8.95? Wait, no, 14 - 5.05 = 8.95? Wait, 14 - 5 = 9, so 14 - 5.05 = 8.95? Wait, no, 5.05 + 8.95 = 14. Yes. Wait, but let's check again. Wait, pOH is 5.05, so pH = 14 - 5.05 = 8.95? Wait, no, that can't be. Wait, no—wait, LiOH is a base, so OH⁻ is in excess, so pH should be above 7. 8.95 is above 7, correct. Wait, but let's recalculate: 14 - 5.05 = 8.95. Yes.
Step2: Confirm with the options
The options include 8.95, which matches 14 - 5.05 = 8.95.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
5.05 (corresponding to the option "5.05")