QUESTION IMAGE
Question
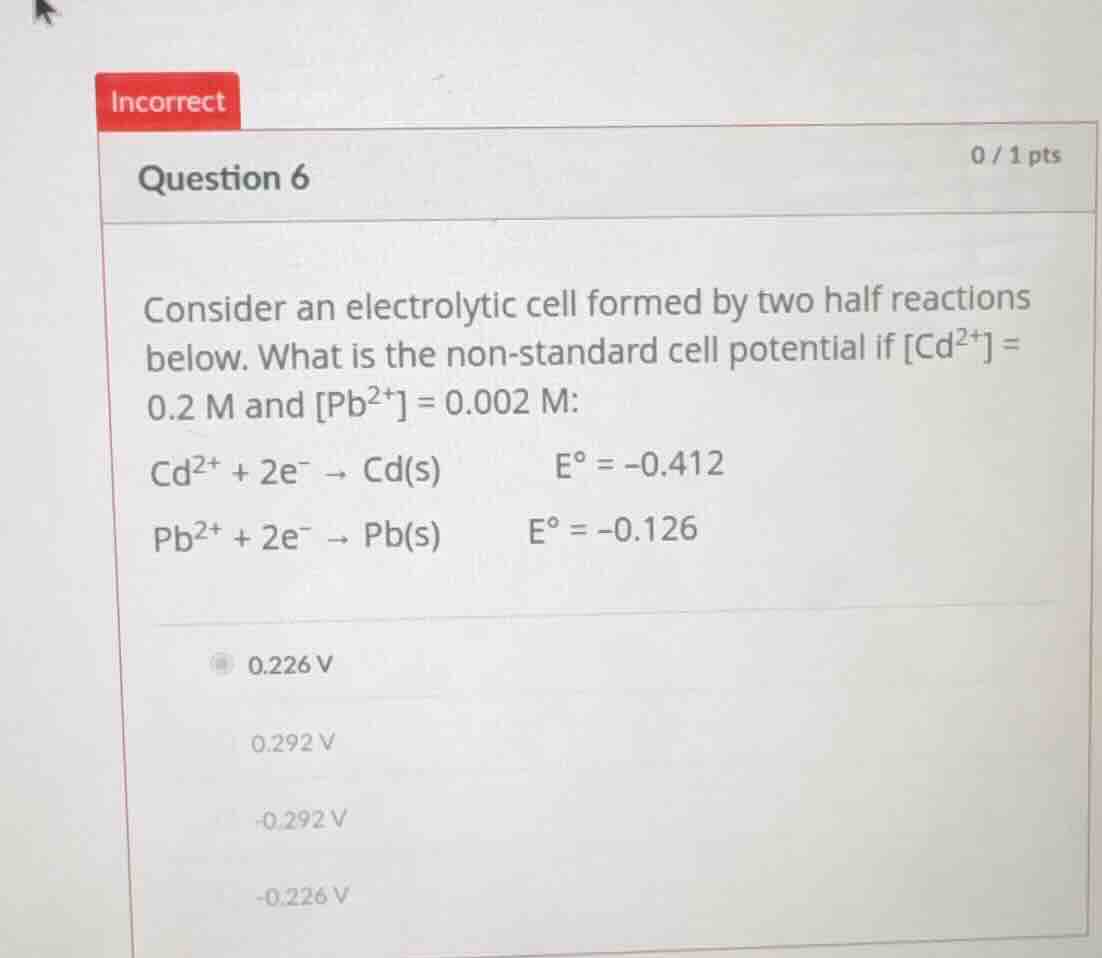
question 6
consider an electrolytic cell formed by two half reactions below. what is the non - standard cell potential if cd²⁺ = 0.2 m and pb²⁺ = 0.002 m:
cd²⁺ + 2e⁻ → cd(s) e° = - 0.412
pb²⁺ + 2e⁻ → pb(s) e° = - 0.126
0.226 v
0.292 v
- 0.292 v
- 0.226 v
Step1: Identify Anode and Cathode
In an electrolytic cell, the non - spontaneous reaction occurs, so the anode (oxidation) is the half - reaction with the more negative \(E^{\circ}\), and the cathode (reduction) is the half - reaction with the less negative \(E^{\circ}\).
For \(Cd^{2+}+2e^{-}
ightarrow Cd(s)\), \(E^{\circ}_{red}=- 0.412\ V\)
For \(Pb^{2+}+2e^{-}
ightarrow Pb(s)\), \(E^{\circ}_{red}=-0.126\ V\)
Since \(-0.412<-0.126\), \(Cd\) is oxidized (anode: \(Cd(s)
ightarrow Cd^{2+}(aq)+2e^{-}\), \(E^{\circ}_{ox} = 0.412\ V\)) and \(Pb^{2+}\) is reduced (cathode: \(Pb^{2+}(aq)+2e^{-}
ightarrow Pb(s)\), \(E^{\circ}_{red}=-0.126\ V\))
The standard cell potential \(E^{\circ}_{cell}=E^{\circ}_{cathode}-E^{\circ}_{anode}\) (or \(E^{\circ}_{red,cathode}+E^{\circ}_{ox,anode}\))
\(E^{\circ}_{cell}=- 0.126+0.412 = 0.286\ V\)
Step2: Use Nernst Equation
The Nernst equation is \(E_{cell}=E^{\circ}_{cell}-\frac{RT}{nF}\ln Q\). At \(298\ K\), \(\frac{RT}{F}\ln10 = 0.0592\ V\), so the Nernst equation can be written as \(E_{cell}=E^{\circ}_{cell}-\frac{0.0592}{n}\log Q\)
For the reaction \(Cd(s)+Pb^{2+}(aq)
ightarrow Cd^{2+}(aq)+Pb(s)\), \(n = 2\) (number of moles of electrons transferred)
The reaction quotient \(Q=\frac{[Cd^{2+}]}{[Pb^{2+}]}\) (since solids are not included in \(Q\))
Given \([Cd^{2+}]=0.2\ M\) and \([Pb^{2+}]=0.002\ M\)
\(Q=\frac{0.2}{0.002}=100\)
Step3: Calculate \(E_{cell}\)
Substitute into the Nernst equation:
\(E_{cell}=E^{\circ}_{cell}-\frac{0.0592}{n}\log Q\)
\(E^{\circ}_{cell}=0.286\ V\), \(n = 2\), \(Q = 100\)
\(\log Q=\log100 = 2\)
\(E_{cell}=0.286-\frac{0.0592}{2}\times2\)
\(E_{cell}=0.286 - 0.0592\)
\(E_{cell}=0.2268\approx0.226\ V\)? Wait, no, wait. Wait, in an electrolytic cell, the cell potential should be negative because the reaction is non - spontaneous (we need to apply external voltage). Wait, I made a mistake in identifying anode and cathode for electrolytic cell.
Wait, in an electrolytic cell, the cell potential \(E_{cell}\) for the non - spontaneous reaction is \(E_{cell}=E^{\circ}_{cell}\) (for spontaneous is positive, for non - spontaneous, \(E^{\circ}_{cell}\) is negative and we need to apply voltage). Wait, let's re - do:
The two half - reactions:
Reduction: \(Pb^{2+}+2e^{-}
ightarrow Pb\), \(E^{\circ}_{red}=-0.126\ V\)
Reduction: \(Cd^{2+}+2e^{-}
ightarrow Cd\), \(E^{\circ}_{red}=-0.412\ V\)
For electrolytic cell, the reaction that occurs is the non - spontaneous one. So we need to reverse the more spontaneous reaction (the one with less negative \(E^{\circ}_{red}\) is more spontaneous as a reduction). So the spontaneous reaction (galvanic) would be \(Cd
ightarrow Cd^{2+}+2e^{-}\) (oxidation, \(E^{\circ}_{ox}=0.412\)) and \(Pb^{2+}+2e^{-}
ightarrow Pb\) (reduction, \(E^{\circ}_{red}=-0.126\)), so galvanic \(E^{\circ}_{cell}=0.412 - 0.126 = 0.286\ V\) (spontaneous). But in electrolytic cell, we are forcing the reverse reaction: \(Cd^{2+}+Pb
ightarrow Cd + Pb^{2+}\), so the cell reaction is \(Cd^{2+}(aq)+Pb(s)
ightarrow Cd(s)+Pb^{2+}(aq)\)
So for this electrolytic reaction, the half - reactions:
Anode (oxidation): \(Pb(s)
ightarrow Pb^{2+}(aq)+2e^{-}\), \(E^{\circ}_{ox}=-(-0.126)=0.126\ V\) (since reverse of \(Pb^{2+}+2e^{-}
ightarrow Pb\) is oxidation, \(E^{\circ}_{ox}=-E^{\circ}_{red}\))
Cathode (reduction): \(Cd^{2+}(aq)+2e^{-}
ightarrow Cd(s)\), \(E^{\circ}_{red}=-0.412\ V\)
\(E^{\circ}_{cell}=E^{\circ}_{cathode}+E^{\circ}_{anode}=-0.412 + 0.126=-0.286\ V\)
Now, the Nernst equation for this reaction: \(Cd^{2+}(aq)+Pb(s)
ightarrow Cd(s)+Pb^{2+}(aq)\)
\(n = 2\), \(Q=\frac{[Pb^{2+}]}{[…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 0.226 V (corresponding to the option - 0.226 V)