QUESTION IMAGE
Question
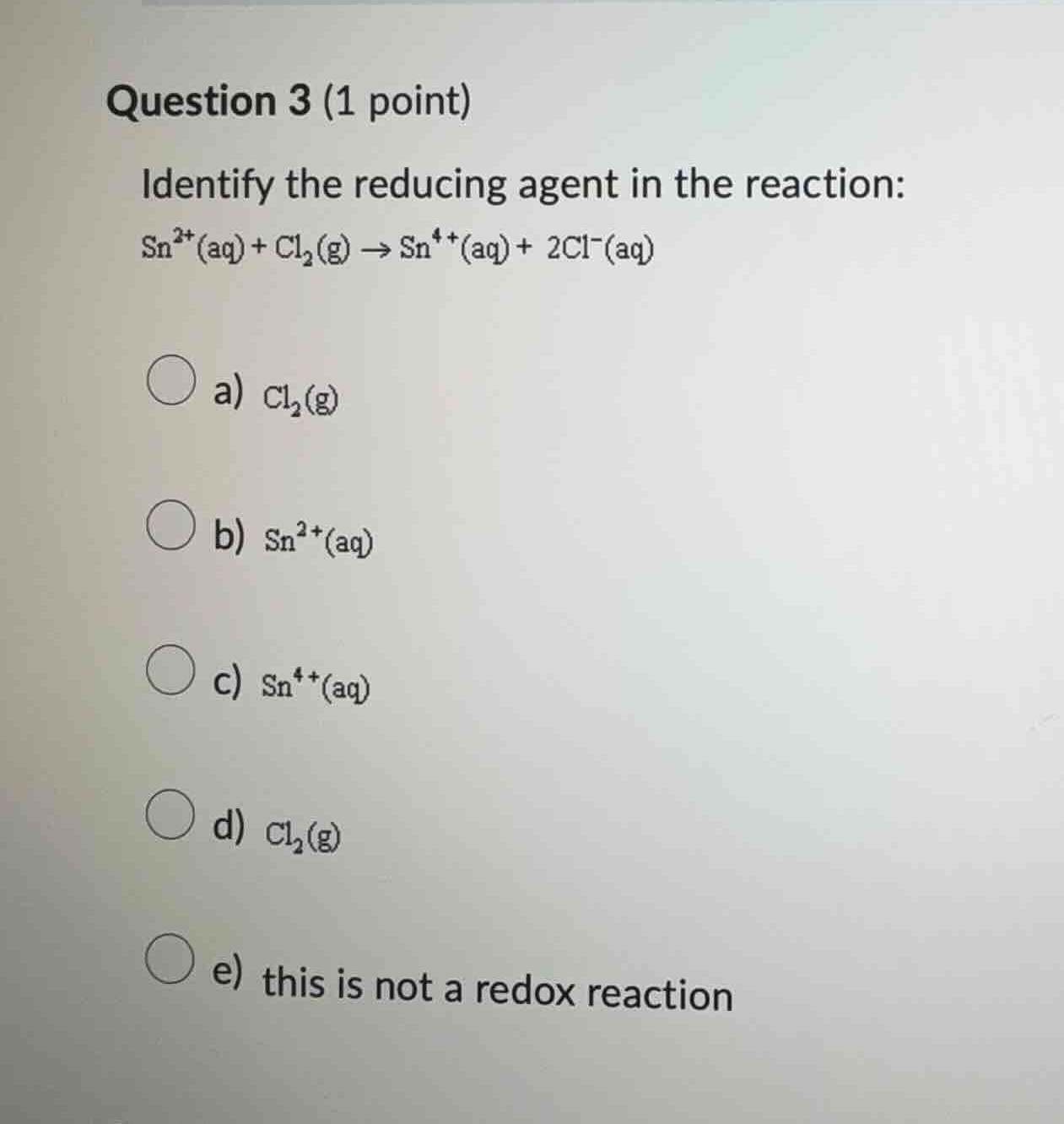
question 3 (1 point)
identify the reducing agent in the reaction:
$\ce{sn^{2+}(aq) + cl_{2}(g) \
ightarrow sn^{4+}(aq) + 2cl^{-}(aq)}$
\\(\bigcirc\\) a) \\(\ce{cl_{2}(g)}
\\(\bigcirc\\) b) \\(\ce{sn^{2+}(aq)}
\\(\bigcirc\\) c) \\(\ce{sn^{4+}(aq)}
\\(\bigcirc\\) d) \\(\ce{cl_{2}(g)}
\\(\bigcirc\\) e) this is not a redox reaction
To identify the reducing agent, we analyze the oxidation states. In \( \text{Sn}^{2+}(\text{aq}) \), Sn has an oxidation state of +2. In \( \text{Sn}^{4+}(\text{aq}) \), Sn has an oxidation state of +4, so Sn is oxidized (loses electrons). A reducing agent is oxidized in a redox reaction. For \( \text{Cl}_2(\text{g}) \), Cl has an oxidation state of 0, and in \( \text{Cl}^-(\text{aq}) \), it is -1, so Cl is reduced (gains electrons), meaning \( \text{Cl}_2 \) is the oxidizing agent. Since \( \text{Sn}^{2+} \) is oxidized, it is the reducing agent. Option e is incorrect because there is a change in oxidation states (redox reaction). Options a and d (same as a) are oxidizing agents, and c is a product, not a reactant acting as a reducing agent.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b) \( \text{Sn}^{2+}(\text{aq}) \)