QUESTION IMAGE
Question
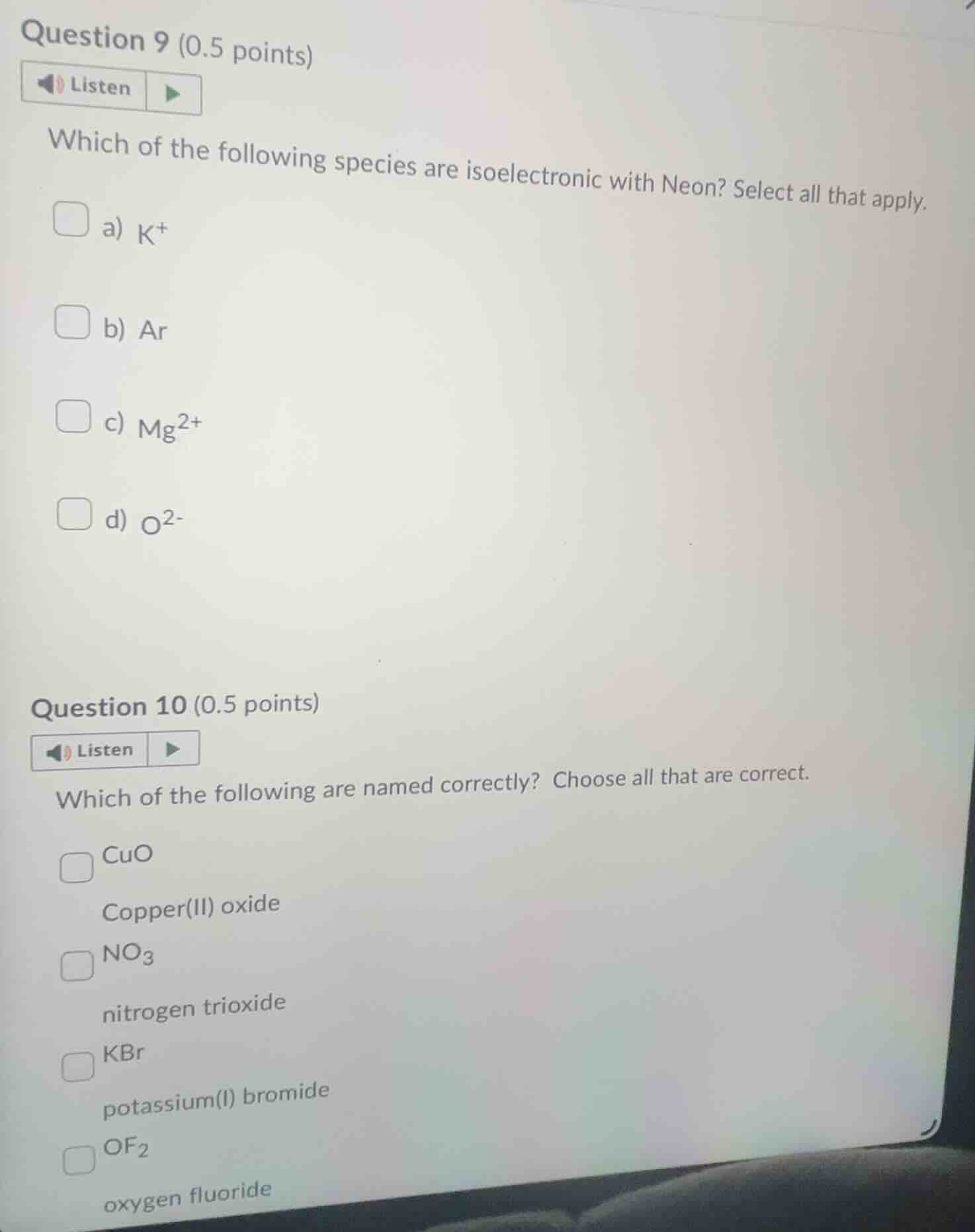
question 9 (0.5 points)listenwhich of the following species are isoelectronic with neon? select all that apply.a) $k^+$b) arc) $mg^{2+}$d) $o^{2-}$question 10 (0.5 points)listenwhich of the following are named correctly? choose all that are correct.cuocopper(ii) oxide$no_3$nitrogen trioxidekbrpotassium(i) bromide$of_2$oxygen fluoride
Step1: Find Ne's electron count
Neon (Ne) has an atomic number of 10, so it has 10 electrons.
Step2: Calculate electrons for each species (Q9)
a) $\text{K}^+$: K has atomic number 19, loses 1 electron: $19-1=18$ electrons
b) $\text{Ar}$: Atomic number 18, has 18 electrons
c) $\text{Mg}^{2+}$: Mg has atomic number 12, loses 2 electrons: $12-2=10$ electrons
d) $\text{O}^{2-}$: O has atomic number 8, gains 2 electrons: $8+2=10$ electrons
Step3: Check correct chemical names (Q10)
- $\text{CuO}$: Cu has +2 charge, so name is Copper(II) oxide (correct)
- $\text{NO}_3$: This is the nitrate ion, nitrogen trioxide is $\text{N}_2\text{O}_3$ (incorrect)
- $\text{KBr}$: K only has +1 charge, so name is potassium bromide (no (I), incorrect)
- $\text{OF}_2$: The correct name is oxygen difluoride (missing "di-", incorrect)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 9
c) $\text{Mg}^{2+}$
d) $\text{O}^{2-}$
Question 10
CuO: Copper(II) oxide