QUESTION IMAGE
Question
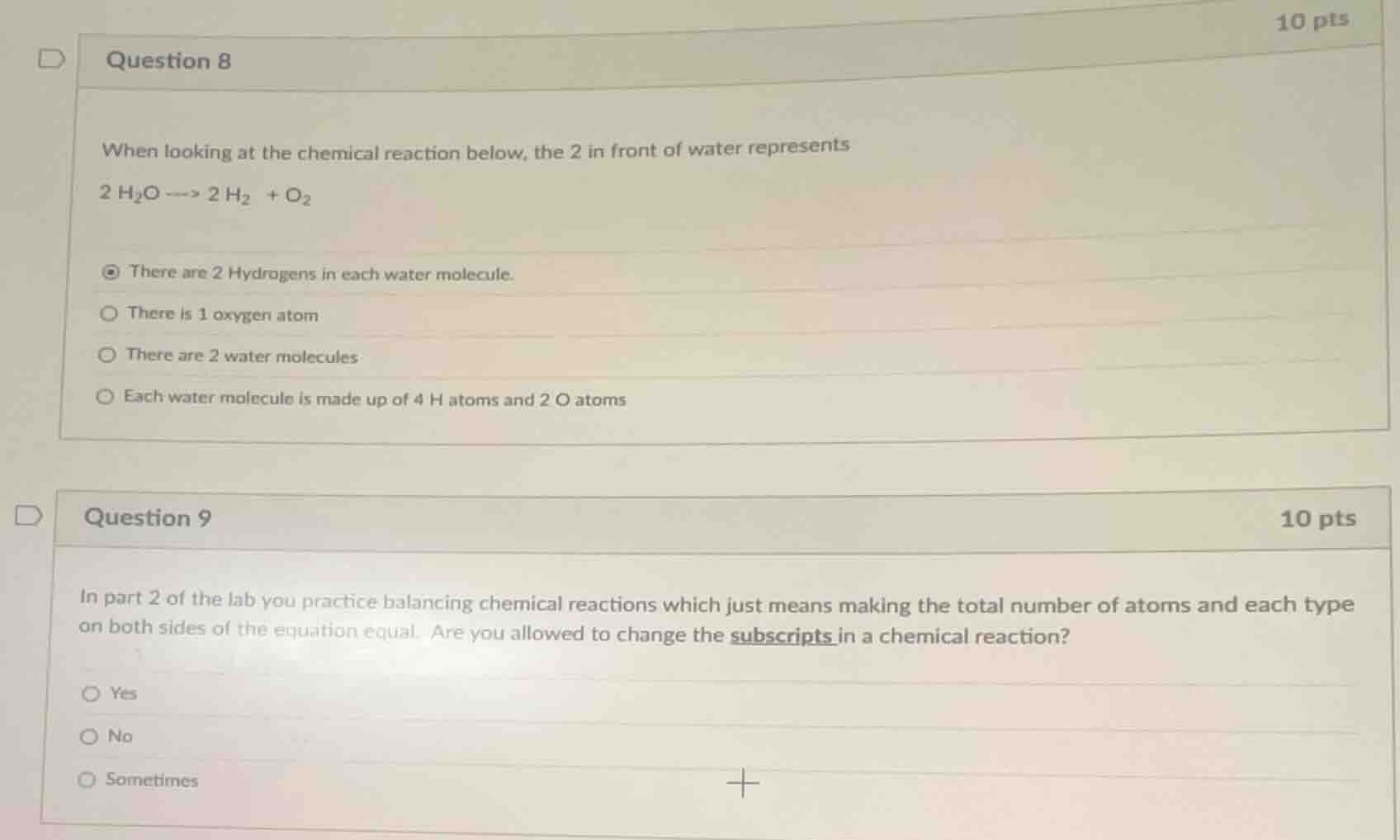
question 8
when looking at the chemical reaction below, the 2 in front of water represents
2 h₂o ----> 2 h₂ + o₂
there are 2 hydrogens in each water molecule.
there is 1 oxygen atom
there are 2 water molecules
each water molecule is made up of 4 h atoms and 2 o atoms
question 9
10 pts
in part 2 of the lab you practice balancing chemical reactions which just means making the total number of atoms and each type on both sides of the equation equal. are you allowed to change the subscripts in a chemical reaction?
yes
no
sometimes
For Question 8: In chemical equations, coefficients (numbers in front of formulas) represent the number of molecules/moles of the substance. Subscripts (small numbers after elements) represent atoms per molecule. The 2 in front of $\text{H}_2\text{O}$ is a coefficient, so it means 2 water molecules.
For Question 9: Balancing equations uses coefficients to adjust atom counts. Changing subscripts would alter the identity of the chemical substance, which is not allowed.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 8: There are 2 water molecules
Question 9: No