QUESTION IMAGE
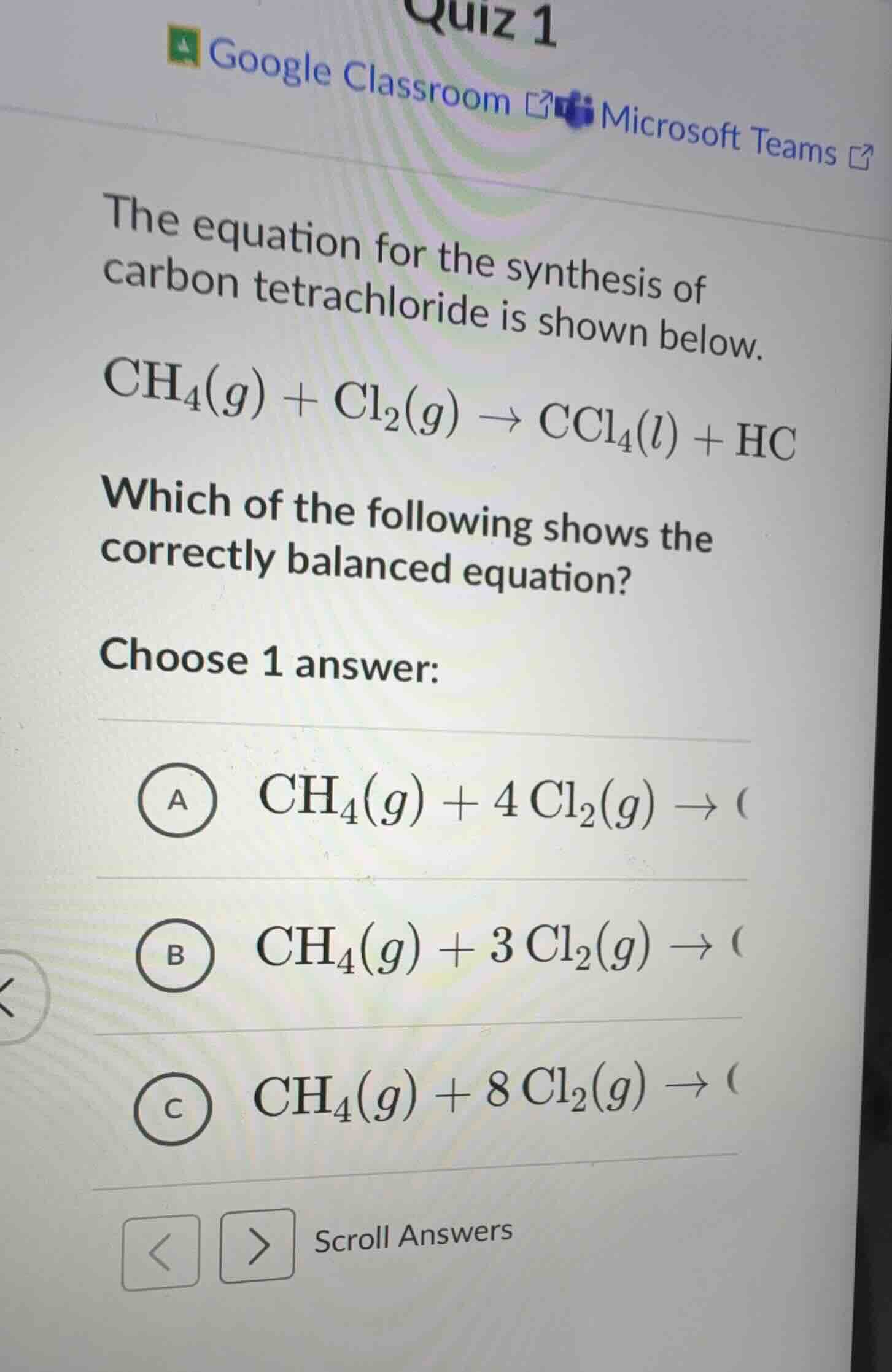
Question
quiz 1
google classroom microsoft teams
the equation for the synthesis of carbon tetrachloride is shown below.
ch₄(g) + cl₂(g) → ccl₄(l) + hc
which of the following shows the correctly balanced equation?
choose 1 answer:
a ch₄(g) + 4 cl₂(g) → (
b ch₄(g) + 3 cl₂(g) → (
c ch₄(g) + 8 cl₂(g) → (
scroll answers
Step1: Identify reactants and products
Reactants: $\ce{CH4(g)}$, $\ce{Cl2(g)}$; Products: $\ce{CCl4(l)}$, $\ce{HCl}$ (assuming the incomplete product is $\ce{HCl}$ as it's a chlorination reaction).
Step2: Balance Carbon
Carbon atoms: 1 in $\ce{CH4}$ and 1 in $\ce{CCl4}$, so C is balanced.
Step3: Balance Hydrogen
Hydrogen atoms: 4 in $\ce{CH4}$, so we need 4 $\ce{HCl}$ (since each $\ce{HCl}$ has 1 H).
Step4: Balance Chlorine
Chlorine in $\ce{HCl}$: 4, and in $\ce{CCl4}$: 4, total Cl: 8. Each $\ce{Cl2}$ has 2 Cl, so number of $\ce{Cl2}$ needed: $\frac{8}{2} = 4$.
So the balanced equation is $\ce{CH4(g) + 4Cl2(g) -> CCl4(l) + 4HCl}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. $\ce{CH4(g) + 4Cl2(g) -> CCl4(l) + 4HCl}$ (assuming the full product for option A is as derived)