QUESTION IMAGE
Question
quiz instructions
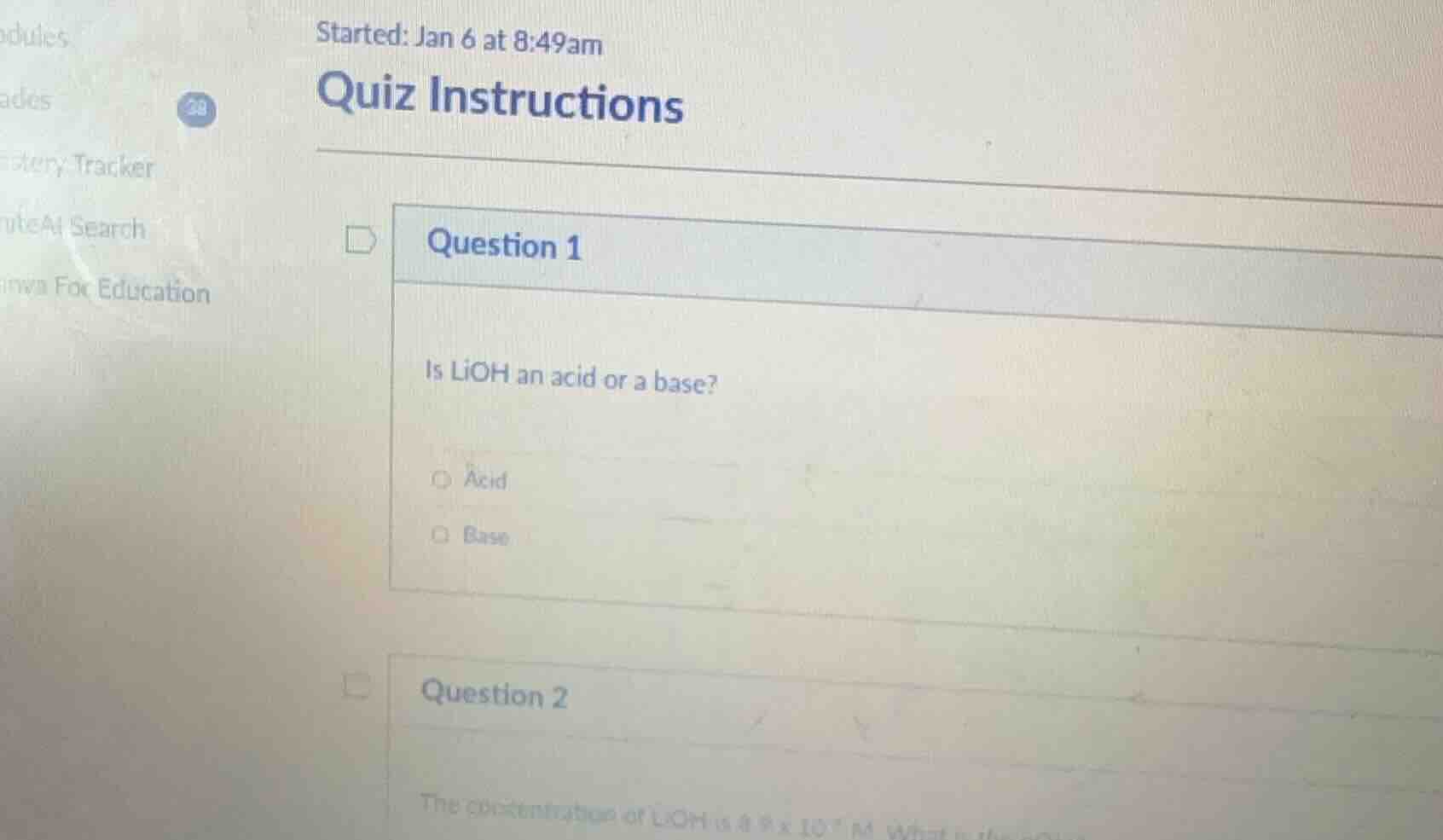
question 1
is lioh an acid or a base?
acid
base
question 2
the concentration of lioh is 4.9×10⁻⁷ m...
Brief Explanations
LiOH is lithium hydroxide. According to the definition of bases (Arrhenius base: a substance that produces hydroxide ions ($\ce{OH^-}$) in aqueous solution), LiOH dissociates in water as $\ce{LiOH -> Li^+ + OH^-}$, producing hydroxide ions. So it is a base.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Base