QUESTION IMAGE
Question
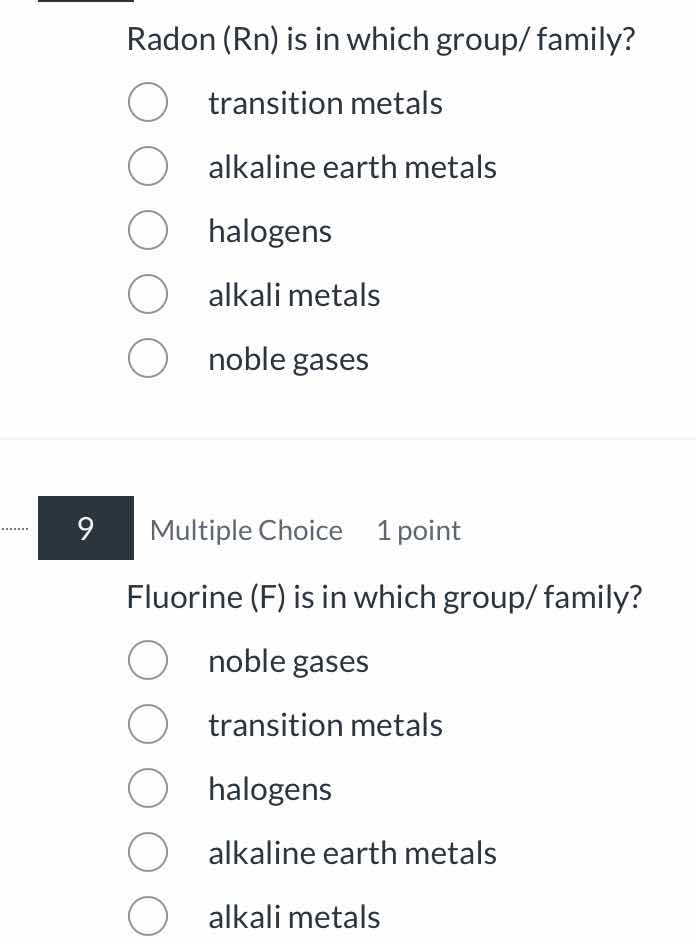
radon (rn) is in which group/ family?
transition metals
alkaline earth metals
halogens
alkali metals
noble gases
9 multiple choice 1 point
fluorine (f) is in which group/ family?
noble gases
transition metals
halogens
alkaline earth metals
alkali metals
Brief Explanations
Radon (Rn) is an inert, non-reactive element with a full valence electron shell, which defines noble gases. Fluorine (F) is a highly reactive nonmetal with 7 valence electrons, a key characteristic of halogens.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- noble gases
- halogens