QUESTION IMAGE
Question
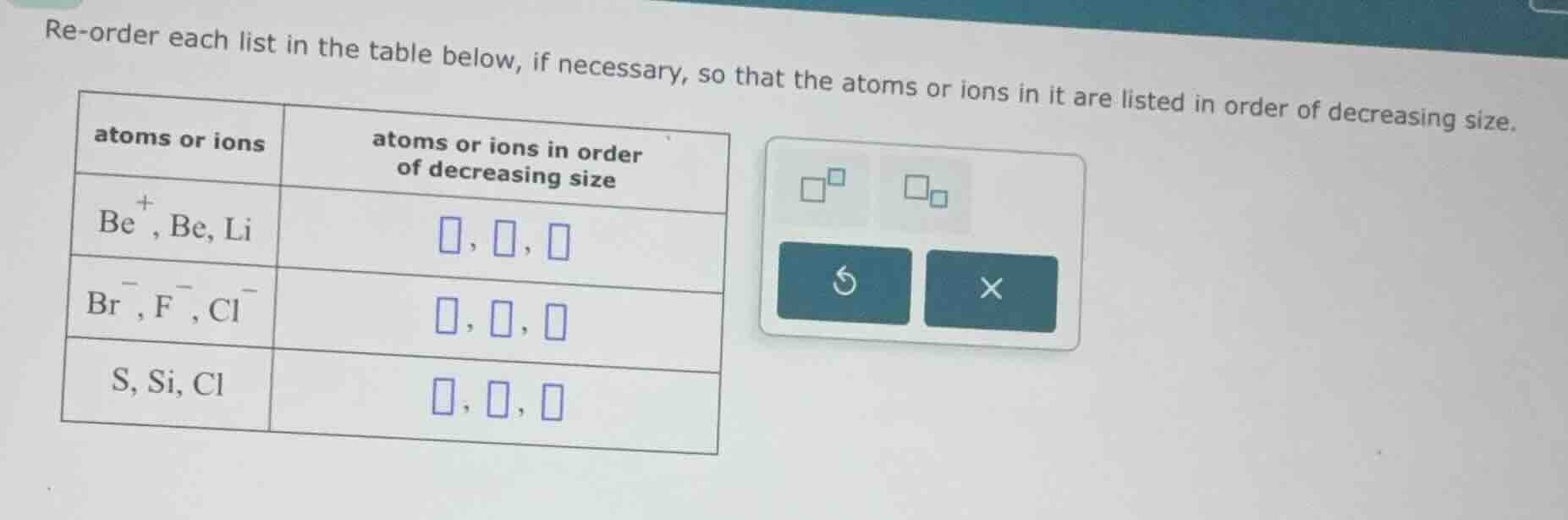
re-order each list in the table below, if necessary, so that the atoms or ions in it are listed in order of decreasing size.
atoms or ions\tatoms or ions in order of decreasing size
be⁺, be, li\t□, □, □
br⁻, f⁻, cl⁻\t□, □, □
s, si, cl\t□, □, □
Step1: Analyze first set (Li, Be, Be⁺)
Atomic size: Li > Be; cation size < parent atom, so Be > Be⁺. Order: $\text{Li} > \text{Be} > \text{Be}^+$
Step2: Analyze second set (Br⁻, Cl⁻, F⁻)
For halide ions, size increases down group. Order: $\text{Br}^- > \text{Cl}^- > \text{F}^-$
Step3: Analyze third set (Si, S, Cl)
Across period, size decreases. Order: $\text{Si} > \text{S} > \text{Cl}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| atoms or ions | atoms or ions in order of decreasing size |
|---|---|
| $\text{Br}^-, \text{F}^-, \text{Cl}^-$ | $\text{Br}^-, \text{Cl}^-, \text{F}^-$ |
| $\text{S}, \text{Si}, \text{Cl}$ | $\text{Si}, \text{S}, \text{Cl}$ |