QUESTION IMAGE
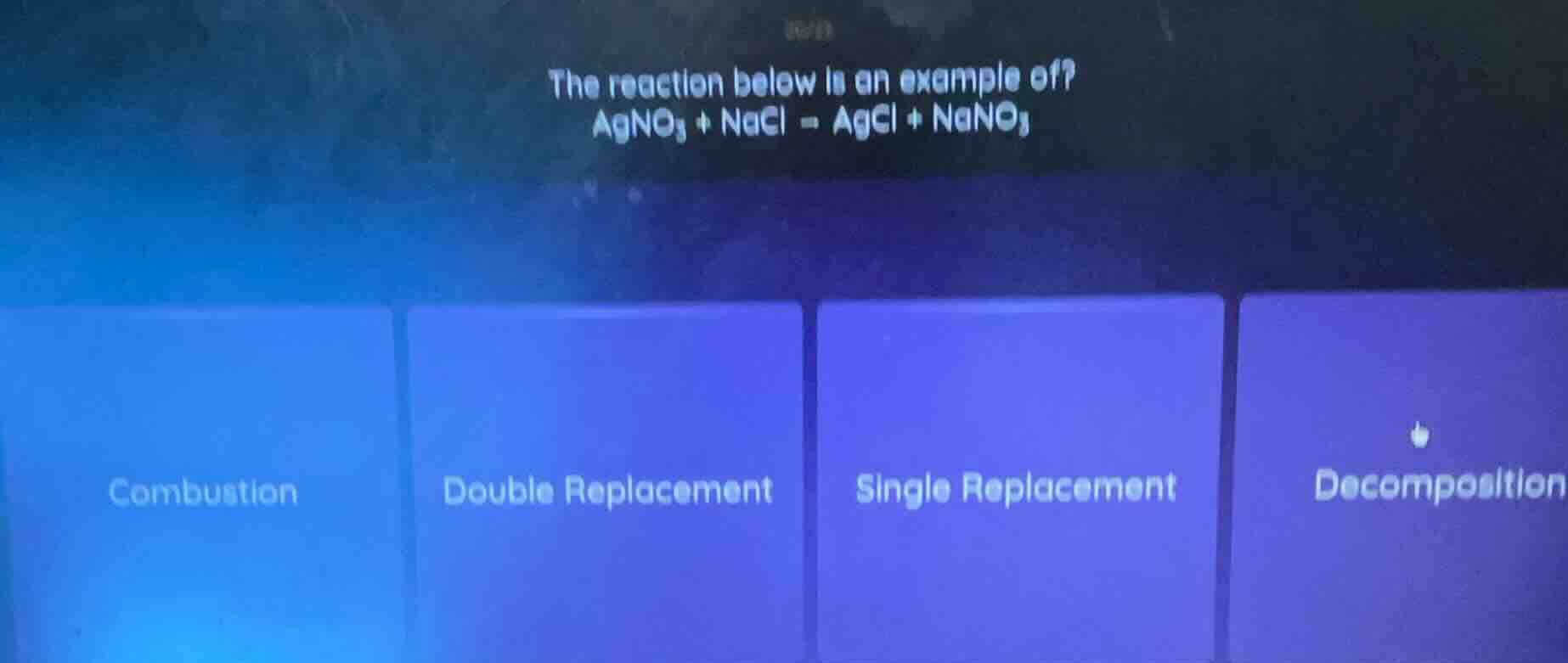
Question
the reaction below is an example of?
$ce{agno_{3} + nacl = agcl + nano_{3}}$
options: combustion, double replacement, single replacement, decomposition
In the reaction $\ce{AgNO_{3} + NaCl = AgCl + NaNO_{3}}$, the cations ($\ce{Ag+}$ and $\ce{Na+}$) and anions ($\ce{NO^{-}_{3}}$ and $\ce{Cl-}$) of the two reactants exchange places to form two new compounds. This matches the definition of a double - replacement reaction. Combustion involves reaction with oxygen, single - replacement has one element replacing another in a compound, and decomposition is breaking a compound into simpler substances, none of which fit here.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. Double Replacement