QUESTION IMAGE
Question
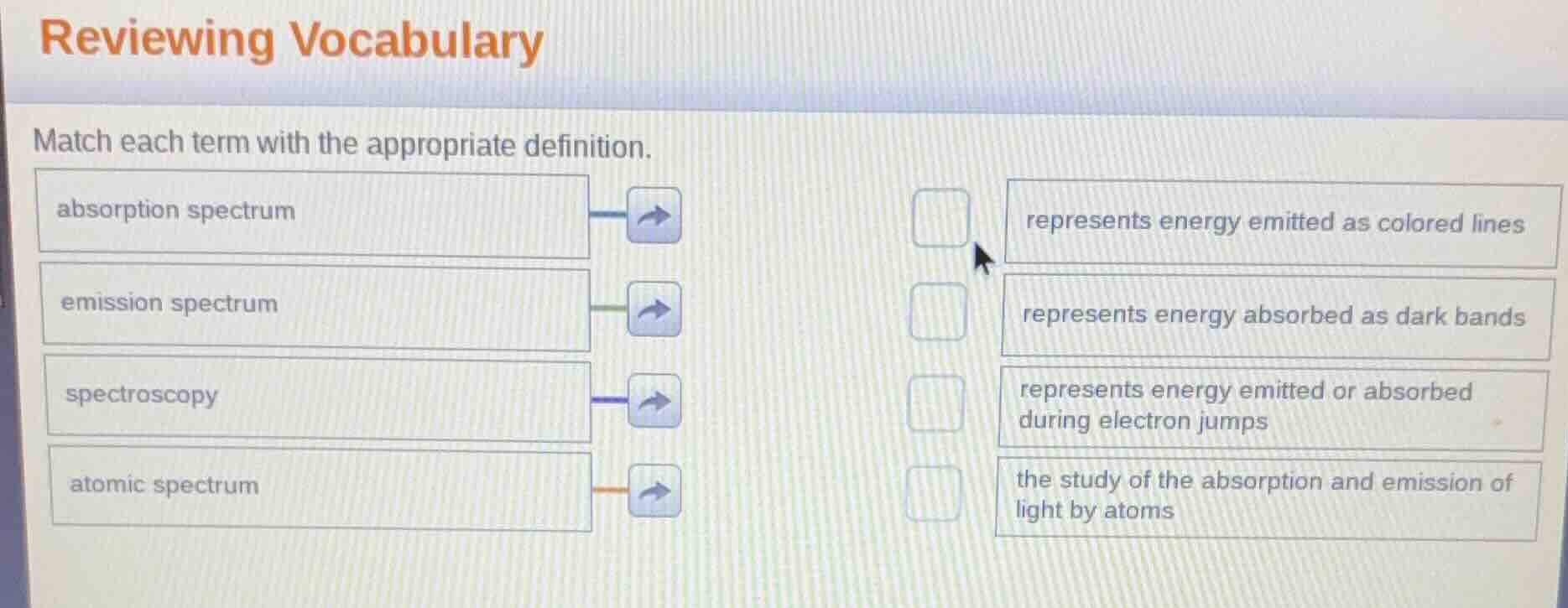
reviewing vocabulary
match each term with the appropriate definition.
absorption spectrum
emission spectrum
spectroscopy
atomic spectrum
represents energy emitted as colored lines
represents energy absorbed as dark bands
represents energy emitted or absorbed during electron jumps
the study of the absorption and emission of light by atoms
Brief Explanations
- An absorption spectrum shows dark bands where specific wavelengths of light are absorbed by a substance.
- An emission spectrum appears as bright colored lines when a substance emits energy.
- Spectroscopy is the scientific field focused on studying how atoms absorb and emit light.
- An atomic spectrum encompasses the energy changes (emitted or absorbed) that occur when electrons move between energy levels in atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- absorption spectrum: represents energy absorbed as dark bands
- emission spectrum: represents energy emitted as colored lines
- spectroscopy: the study of the absorption and emission of light by atoms
- atomic spectrum: represents energy emitted or absorbed during electron jumps