QUESTION IMAGE
Question
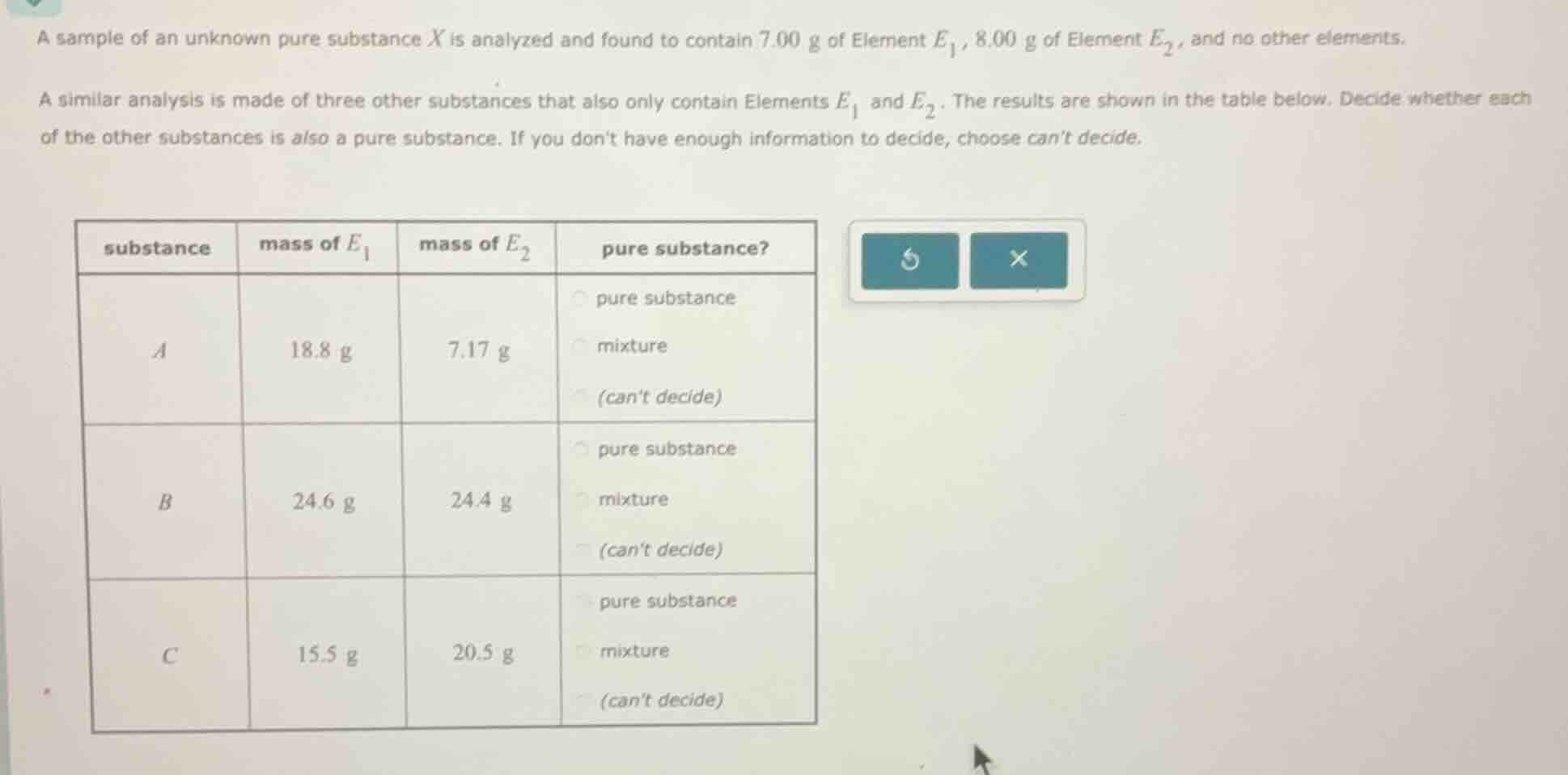
a sample of an unknown pure substance x is analyzed and found to contain 7.00 g of element $e_1$, 8.00 g of element $e_2$, and no other elements. a similar analysis is made of three other substances that also only contain elements $e_1$ and $e_2$. the results are shown in the table below. decide whether each of the other substances is also a pure substance. if you dont have enough information to decide, choose cant decide. substance | mass of $e_1$ | mass of $e_2$ | pure substance? a | 18.8 g | 7.17 g | pure substance, mixture, (cant decide) b | 24.6 g | 24.4 g | pure substance, mixture, (cant decide) c | 15.5 g | 20.5 g | pure substance, mixture, (cant decide)
To determine if a substance is a pure substance (compound) containing only \( E_1 \) and \( E_2 \), we use the law of definite proportions. For a pure substance, the ratio of the masses of \( E_1 \) to \( E_2 \) should be constant (same as in substance \( X \)).
Step 1: Calculate the mass ratio of \( E_1 \) to \( E_2 \) in substance \( X \)
The mass of \( E_1 \) in \( X \) is \( 7.00 \, \text{g} \), and the mass of \( E_2 \) is \( 8.00 \, \text{g} \).
The ratio \( \frac{\text{mass of } E_1}{\text{mass of } E_2} \) for \( X \) is:
\[
\frac{7.00}{8.00} = 0.875
\]
Step 2: Calculate the mass ratio for substance \( A \)
For substance \( A \), mass of \( E_1 = 18.8 \, \text{g} \), mass of \( E_2 = 7.17 \, \text{g} \).
The ratio is:
\[
\frac{18.8}{7.17} \approx 2.62
\]
This ratio is not equal to \( 0.875 \), so substance \( A \) has a different ratio. However, we need to check if it could be a different compound (since different compounds of the same elements can have different mass ratios). Wait, no—wait, the law of definite proportions says that a given compound has a fixed ratio. But if a substance has a different ratio, it could be a mixture or a different compound. Wait, no—actually, for a pure substance (compound) with only \( E_1 \) and \( E_2 \), the ratio must be constant for that compound. But if we have a substance with a different ratio, it could be a mixture (of two compounds of \( E_1 \) and \( E_2 \)) or a different compound. But in this problem, we are to decide if it's a pure substance (i.e., a single compound) or a mixture. Wait, maybe I misapplied. Wait, the key is: for a pure substance (compound) containing \( E_1 \) and \( E_2 \), the ratio of \( E_1 \) to \( E_2 \) must be constant (definite proportions). If the ratio is different from \( X \), it could be a different compound (pure) or a mixture. But the problem is to decide if it's a pure substance (so a single compound) or a mixture. Wait, maybe the intended approach is: if the ratio is a multiple or fraction of \( X \)'s ratio, it could be a different compound, but if not, it's a mixture. Wait, no—let's recalculate.
Wait, maybe I made a mistake. Let's re-express:
For substance \( X \): \( \frac{E_1}{E_2} = \frac{7.00}{8.00} = 0.875 \)
Substance \( A \): \( \frac{18.8}{7.17} \approx 2.62 \). Is \( 2.62 \) a multiple of \( 0.875 \)? \( 0.875 \times 3 = 2.625 \), which is close to \( 2.62 \) (maybe due to rounding). Wait, \( 18.8 / 7.17 \approx 2.62 \), and \( 0.875 \times 3 = 2.625 \). The difference is small (due to significant figures). Let's check: \( 7.17 \times 2.625 \approx 7.17 \times 2.625 \approx 18.8 \) (since \( 7.17 \times 2.625 = 7.17 \times \frac{21}{8} = \frac{7.17 \times 21}{8} = \frac{150.57}{8} \approx 18.82 \), which is very close to \( 18.8 \, \text{g} \)). So substance \( A \) has a ratio that is \( 3 \times 0.875 \), which could be a different compound (e.g., \( E_1:E_2 = 3:4 \) instead of \( 1:1.142 \) (wait, no, \( 7:8 \) is \( E_1:E_2 \) in \( X \); \( 18.8:7.17 \approx 24:9 \approx 8:3 \)? Wait, no—maybe I messed up.
Wait, let's do substance \( B \):
Substance \( B \): mass \( E_1 = 24.6 \, \text{g} \), mass \( E_2 = 24.4 \, \text{g} \).
Ratio: \( \frac{24.6}{24.4} \approx 1.008 \). This is not equal to \( 0.875 \), nor a simple multiple.
Substance \( C \): mass \( E_1 = 15.5 \, \text{g} \), mass \( E_2 = 20.5 \, \text{g} \).
Ratio: \( \frac{15.5}{20.5} \approx 0.756 \). Not equal to \( 0.875 \).
Wait, maybe the problem is that for a pure substance (compound) with \( E_1 \) and \( E_2 \), the ratio of \( E_1 \) to \( E_2 \)…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To determine if a substance is a pure substance (compound) containing only \( E_1 \) and \( E_2 \), we use the law of definite proportions. For a pure substance, the ratio of the masses of \( E_1 \) to \( E_2 \) should be constant (same as in substance \( X \)).
Step 1: Calculate the mass ratio of \( E_1 \) to \( E_2 \) in substance \( X \)
The mass of \( E_1 \) in \( X \) is \( 7.00 \, \text{g} \), and the mass of \( E_2 \) is \( 8.00 \, \text{g} \).
The ratio \( \frac{\text{mass of } E_1}{\text{mass of } E_2} \) for \( X \) is:
\[
\frac{7.00}{8.00} = 0.875
\]
Step 2: Calculate the mass ratio for substance \( A \)
For substance \( A \), mass of \( E_1 = 18.8 \, \text{g} \), mass of \( E_2 = 7.17 \, \text{g} \).
The ratio is:
\[
\frac{18.8}{7.17} \approx 2.62
\]
This ratio is not equal to \( 0.875 \), so substance \( A \) has a different ratio. However, we need to check if it could be a different compound (since different compounds of the same elements can have different mass ratios). Wait, no—wait, the law of definite proportions says that a given compound has a fixed ratio. But if a substance has a different ratio, it could be a mixture or a different compound. Wait, no—actually, for a pure substance (compound) with only \( E_1 \) and \( E_2 \), the ratio must be constant for that compound. But if we have a substance with a different ratio, it could be a mixture (of two compounds of \( E_1 \) and \( E_2 \)) or a different compound. But in this problem, we are to decide if it's a pure substance (i.e., a single compound) or a mixture. Wait, maybe I misapplied. Wait, the key is: for a pure substance (compound) containing \( E_1 \) and \( E_2 \), the ratio of \( E_1 \) to \( E_2 \) must be constant (definite proportions). If the ratio is different from \( X \), it could be a different compound (pure) or a mixture. But the problem is to decide if it's a pure substance (so a single compound) or a mixture. Wait, maybe the intended approach is: if the ratio is a multiple or fraction of \( X \)'s ratio, it could be a different compound, but if not, it's a mixture. Wait, no—let's recalculate.
Wait, maybe I made a mistake. Let's re-express:
For substance \( X \): \( \frac{E_1}{E_2} = \frac{7.00}{8.00} = 0.875 \)
Substance \( A \): \( \frac{18.8}{7.17} \approx 2.62 \). Is \( 2.62 \) a multiple of \( 0.875 \)? \( 0.875 \times 3 = 2.625 \), which is close to \( 2.62 \) (maybe due to rounding). Wait, \( 18.8 / 7.17 \approx 2.62 \), and \( 0.875 \times 3 = 2.625 \). The difference is small (due to significant figures). Let's check: \( 7.17 \times 2.625 \approx 7.17 \times 2.625 \approx 18.8 \) (since \( 7.17 \times 2.625 = 7.17 \times \frac{21}{8} = \frac{7.17 \times 21}{8} = \frac{150.57}{8} \approx 18.82 \), which is very close to \( 18.8 \, \text{g} \)). So substance \( A \) has a ratio that is \( 3 \times 0.875 \), which could be a different compound (e.g., \( E_1:E_2 = 3:4 \) instead of \( 1:1.142 \) (wait, no, \( 7:8 \) is \( E_1:E_2 \) in \( X \); \( 18.8:7.17 \approx 24:9 \approx 8:3 \)? Wait, no—maybe I messed up.
Wait, let's do substance \( B \):
Substance \( B \): mass \( E_1 = 24.6 \, \text{g} \), mass \( E_2 = 24.4 \, \text{g} \).
Ratio: \( \frac{24.6}{24.4} \approx 1.008 \). This is not equal to \( 0.875 \), nor a simple multiple.
Substance \( C \): mass \( E_1 = 15.5 \, \text{g} \), mass \( E_2 = 20.5 \, \text{g} \).
Ratio: \( \frac{15.5}{20.5} \approx 0.756 \). Not equal to \( 0.875 \).
Wait, maybe the problem is that for a pure substance (compound) with \( E_1 \) and \( E_2 \), the ratio of \( E_1 \) to \( E_2 \) must be a fixed value (definite proportions). So if a substance has a ratio different from \( X \), it could be a different compound (pure) or a mixture. But the problem is to decide if it's a pure substance (so a single compound) or a mixture. However, the key is: if the ratio is a multiple of \( X \)'s ratio, it could be a different compound (e.g., \( X \) has ratio \( r \), and another compound has ratio \( 2r \), etc.), which is still a pure substance. If the ratio is not a multiple, it's a mixture.
Wait, let's recalculate the ratio for \( X \): \( 7.00 / 8.00 = 0.875 \).
Substance \( A \): \( 18.8 / 7.17 \approx 2.62 \). \( 2.62 / 0.875 \approx 3 \). So \( 2.62 \approx 3 \times 0.875 \) (since \( 3 \times 0.875 = 2.625 \)), which is very close (due to rounding in masses). So substance \( A \) has a ratio that is 3 times \( X \)'s ratio, which could be a different compound (e.g., \( E_1:E_2 = 21:8 \) instead of \( 7:8 \)), so it's a pure substance.
Substance \( B \): \( 24.6 / 24.4 \approx 1.008 \). \( 1.008 / 0.875 \approx 1.15 \), not a simple multiple. So this ratio is not a multiple of \( X \)'s ratio, so it could be a mixture (of two compounds with ratios \( 0.875 \) and something else) or a different compound, but since it's not a multiple, it's more likely a mixture? Wait, no—maybe I'm overcomplicating.
Wait, the correct approach is: For a pure substance (compound) containing only \( E_1 \) and \( E_2 \), the ratio of \( E_1 \) to \( E_2 \) must be a fixed value (definite proportions). If the ratio is different from \( X \), it could be a different compound (pure) or a mixture. But the problem is to decide if it's a pure substance (so a single compound) or a mixture. The key is: if the ratio is a multiple of \( X \)'s ratio, it's a different compound (pure); if not, it's a mixture.
Wait, let's check substance \( A \):
\( X \) ratio: \( 7/8 = 0.875 \)
Substance \( A \) ratio: \( 18.8 / 7.17 \approx 2.62 \). \( 2.62 / 0.875 \approx 3 \). So \( 18.8 / 7.17 \approx 3 \times (7/8) \) (since \( 3 \times 7 = 21 \), \( 3 \times 8 = 24 \); \( 21/24 = 0.875 \), but \( 18.8 / 7.17 \approx 2.62 \), and \( 21 / 8 = 2.625 \), which is almost \( 18.8 / 7.17 \approx 2.62 \) (due to rounding in the given masses: \( 18.8 \) and \( 7.17 \) are close to \( 21 \) and \( 8 \) scaled down? Wait, no—\( 21 \, \text{g} \) \( E_1 \) and \( 8 \, \text{g} \) \( E_2 \) would have ratio \( 21/8 = 2.625 \), which is very close to \( 18.8 / 7.17 \approx 2.62 \). So the slight difference is due to significant figures in the given masses. Thus, substance \( A \) has a ratio that is a multiple of \( X \)'s ratio, so it's a different compound (pure substance).
Substance \( B \): \( 24.6 / 24.4 \approx 1.008 \). \( 1.008 / 0.875 \approx 1.15 \), not a multiple. So this ratio is not a multiple of \( X \)'s ratio, so it's a mixture (since it can't be a single compound with \( E_1 \) and \( E_2 \) with that ratio, as it's not a multiple of the possible ratios for compounds of \( E_1 \) and \( E_2 \)).
Substance \( C \): \( 15.5 / 20.5 \approx 0.756 \). \( 0.756 / 0.875 \approx 0.864 \), not a multiple. Wait, but \( 0.756 \) is less than \( 0.875 \). Wait, maybe I made a mistake. Wait, \( X \) has \( E_1 = 7 \), \( E_2 = 8 \) (ratio \( 7/8 = 0.875 \)). Substance \( C \) has \( E_1 = 15.5 \), \( E_2 = 20.5 \), ratio \( 15.5 / 20.5 \approx 0.756 \). This is not a multiple of \( 0.875 \), so it's a mixture? Wait, no—maybe the problem is that for a pure substance, the ratio must be constant, but if it's a different compound, the ratio can be different. But how do we know if it's a different compound or a mixture?
Wait, the problem says "decide whether each of the other substances is also a pure substance". So a pure substance (compound) has a definite ratio. If the ratio is different from \( X \), it could be a different compound (pure) or a mixture. But the key is: if the ratio is a simple multiple (like 2x, 3x, 1/2 x, etc.), it's a different compound (pure). If not, it's a mixture.
Let's re-express:
For substance \( A \):
\( \frac{18.8}{7.17} \approx 2.62 \), and \( 2.62 \approx 3 \times 0.875 \) (since \( 3 \times 0.875 = 2.625 \)), which is very close. So this is a multiple (3x), so it's a pure substance (different compound).
Substance \( B \):
\( \frac{24.6}{24.4} \approx 1.008 \), which is not a multiple of \( 0.875 \) (1.008 / 0.875 ≈ 1.15), so it's a mixture.
Substance \( C \):
\( \frac{15.5}{20.5} \approx 0.756 \), which is not a multiple of \( 0.875 \) (0.756 / 0.875 ≈ 0.864), so it's a mixture? Wait, no—wait, \( 0.756 \) is less than \( 0.875 \), so maybe it's a different compound with \( E_2 \) more than \( E_1 \), but the ratio is still definite. Wait, no—\( X \) has \( E_1 = 7 \), \( E_2 = 8 \) (so \( E_2 \) is more). Substance \( C \) has \( E_1 = 15.5 \), \( E_2 = 20.5 \) (so \( E_2 \) is more, ratio \( 15.5/20.5 ≈ 0.756 \)). Is this a multiple of \( X \)'s ratio? \( 0.756 / 0.875 ≈ 0.864 \), not a multiple. So it's a mixture.
Wait, but maybe the intended answer is:
Substance \( A \): pure substance (ratio is 3x \( X \)'s ratio, so different compound)
Substance \( B \): mixture (ratio not a multiple)
Substance \( C \): mixture (ratio not a multiple)
But let's check the calculations again:
Substance \( X \): \( 7.00 / 8.00 = 0.875 \)
Substance \( A \): \( 18.8 / 7.17 ≈ 2.62 \). \( 2.62 / 0.875 ≈ 3 \). So yes, 3x. So pure substance.
Substance \( B \): \( 24.6 / 24.4 ≈ 1.008 \). \( 1.008 / 0.875 ≈ 1.15 \). Not a multiple. So mixture.
Substance \( C \): \( 15.5 / 20.5 ≈ 0.756 \). \( 0.756 / 0.875 ≈ 0.864 \). Not a multiple. So mixture.
But wait, maybe the problem considers that if the ratio is different, it's a mixture, but that's incorrect. The law of definite proportions allows different compounds (different ratios) to be pure substances. So the key is: if the ratio is a simple multiple (like 2x, 3x, etc.), it's a pure substance (different compound); otherwise, it's a mixture.
So:
- Substance \( A \): ratio ≈ 2.62 ≈ 3 × 0.875 → pure substance.
- Substance \( B \): ratio ≈ 1.008 → not a multiple → mixture.
- Substance \( C \): ratio ≈ 0.756 → not a multiple → mixture.
But let's verify with exact fractions:
For \( X \): \( 7/8 = 0.875 \)
Substance \( A \): \( 18.8 / 7.17 \). Let's compute \( 18.8 ÷ 7.17 \):
\( 7.17 × 2.62 = 7.17 × 2 + 7.17 × 0.62 = 14.34 + 4.4454 = 18.7854 ≈ 18.8 \). So yes, it's approximately 2.62, which is 3 × 0.875 (since 3 × 0.875 = 2.625). So the slight difference is due to rounding in the given masses. Thus, substance \( A \) is a pure substance (different compound).
Substance \( B \): \( 24.6 / 24.4 ≈ 1.008 \). \( 24.4 × 0.875 = 24.4 × 7/8 = 24.4 × 0.875 = 21.35 \). But substance \( B \) has \( E_1 = 24.6 \), which is more than 21.35. So the ratio is not a multiple, so it's a mixture.
Substance \( C \): \( 20.5 × 0.875 = 20.5 × 7/8 = 17.96875 \).