QUESTION IMAGE
Question
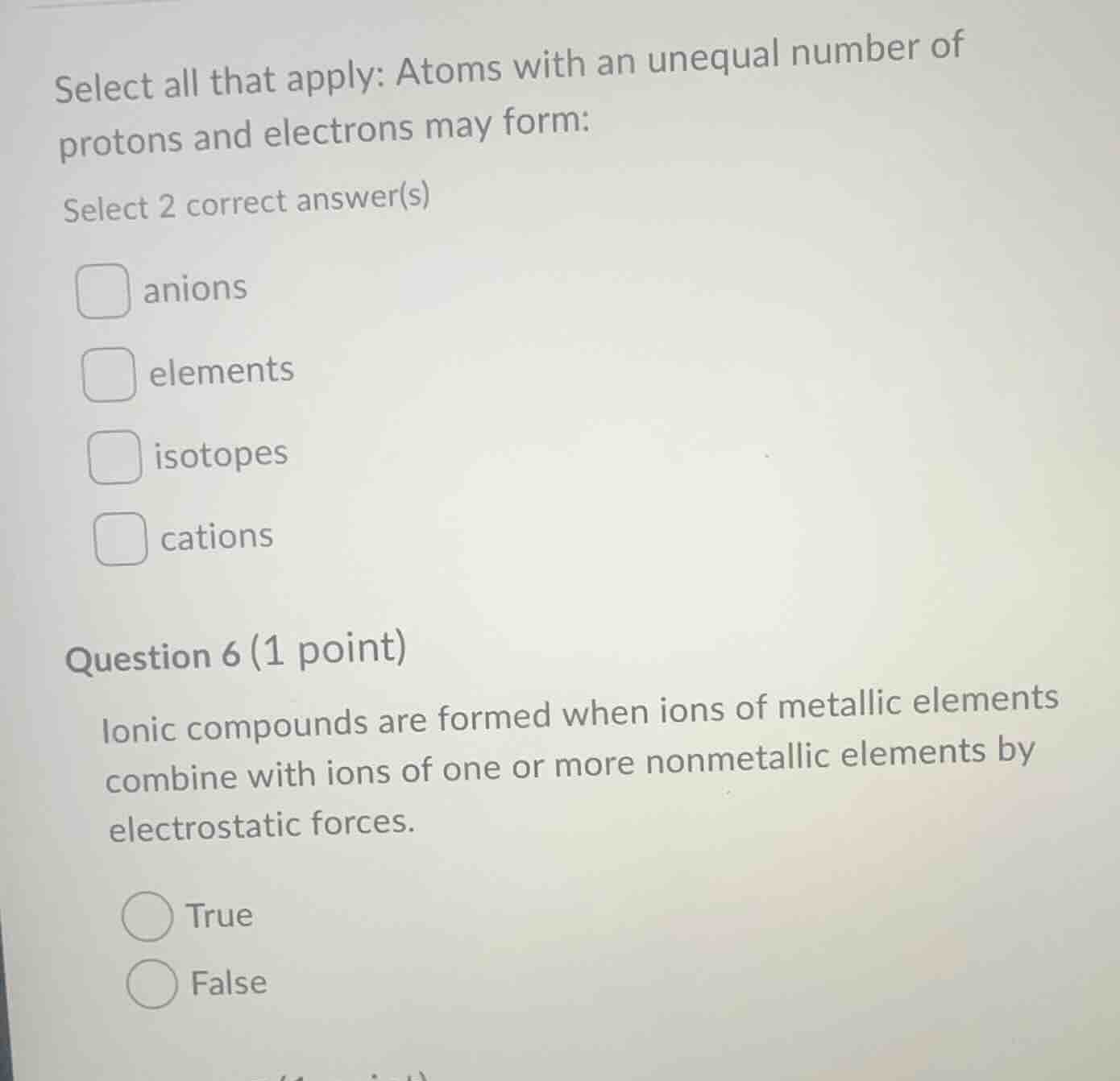
select all that apply: atoms with an unequal number of protons and electrons may form:
select 2 correct answer(s)
□ anions
□ elements
□ isotopes
□ cations
question 6 (1 point)
ionic compounds are formed when ions of metallic elements combine with ions of one or more nonmetallic elements by electrostatic forces.
○ true
○ false
Response
First Question (Select all that apply)
Brief Explanations
- An atom with unequal protons and electrons is an ion. If electrons > protons, it's an anion (negative charge). If protons > electrons, it's a cation (positive charge).
- Elements are neutral (protons = electrons), and isotopes differ in neutrons (protons = electrons in isotopes of the same element). So the correct options are anions and cations.
Brief Explanations
Ionic compounds are formed between metal ions (cations, lose electrons) and non - metal ions (anions, gain electrons) due to electrostatic attraction (ionic bonds) between opposite charges. So the statement is true.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. anions, D. cations