QUESTION IMAGE
Question
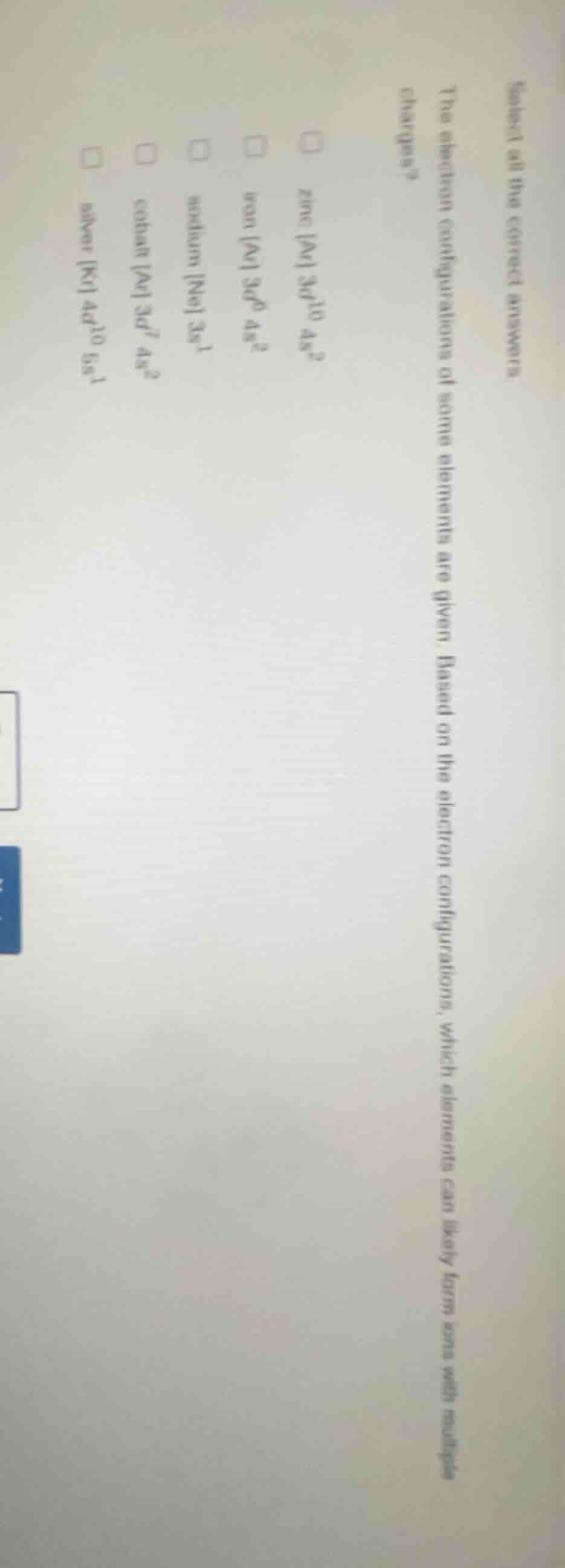
select all the correct answers
the electron configurations of some elements are given. based on the electron configurations, which elements can likely form ions with multiple charges?
□ zinc ar 3d¹⁰ 4s²
□ iron ar 3d⁶ 4s²
□ sodium ne 3s¹
□ cobalt ar 3d⁷ 4s²
□ silver kr 4d¹⁰ 5s¹
To determine which elements can form ions with multiple charges, we analyze their electron configurations:
1. Zinc: $[\text{Ar}] 3d^{10} 4s^2$
Zinc has a filled $3d$ subshell. It typically loses the $4s^2$ electrons to form $\text{Zn}^{2+}$ (only one common charge, $+2$).
2. Iron: $[\text{Ar}] 3d^6 4s^2$
Iron has a partially filled $3d$ subshell. It can lose $2$ electrons (from $4s^2$) to form $\text{Fe}^{2+}$, or $3$ electrons (from $4s^2$ and one $3d$) to form $\text{Fe}^{3+}$. Thus, it can have multiple charges.
3. Sodium: $[\text{Ne}] 3s^1$
Sodium has one valence electron in $3s^1$. It typically loses this electron to form $\text{Na}^+$ (only one common charge, $+1$).
4. Cobalt: $[\text{Ar}] 3d^7 4s^2$
Cobalt has a partially filled $3d$ subshell. It can lose $2$ electrons (from $4s^2$) to form $\text{Co}^{2+}$, or $3$ electrons (from $4s^2$ and one $3d$) to form $\text{Co}^{3+}$. Thus, it can have multiple charges.
5. Silver: $[\text{Kr}] 4d^{10} 5s^1$
Silver has a filled $4d$ subshell. It typically loses the $5s^1$ electron to form $\text{Ag}^+$ (only one common charge, $+1$).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The elements that can form ions with multiple charges are:
- Iron: $[\text{Ar}] 3d^6 4s^2$
- Cobalt: $[\text{Ar}] 3d^7 4s^2$
(Select the checkboxes for iron and cobalt.)