QUESTION IMAGE
Question
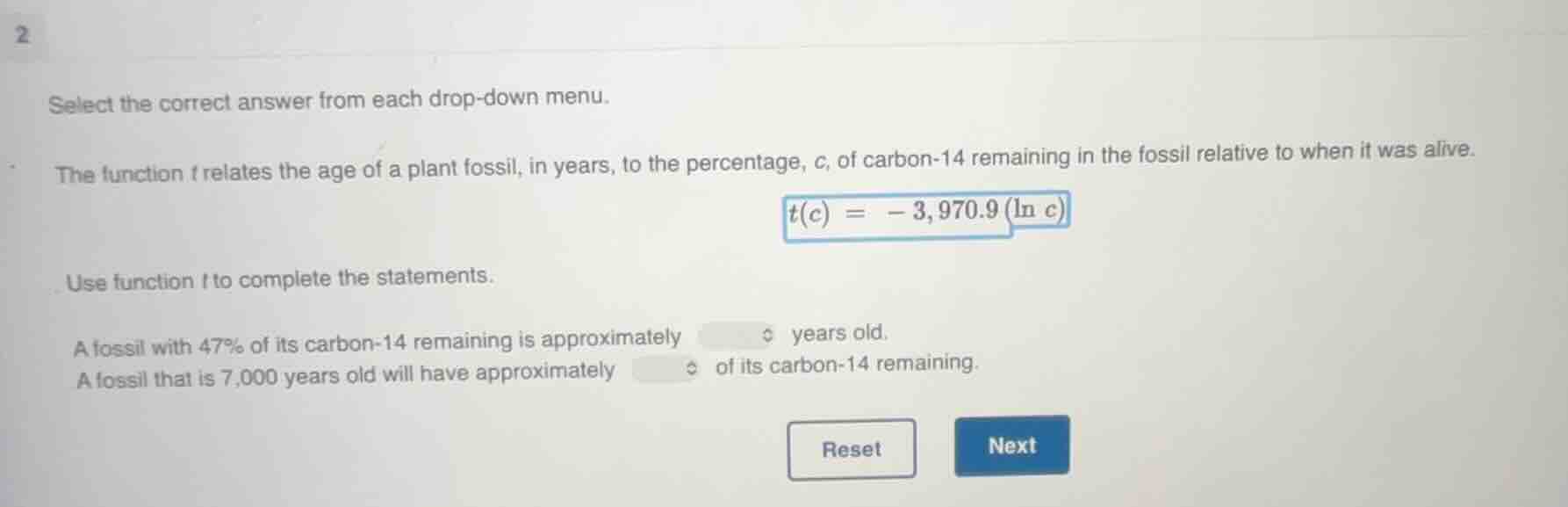
2
select the correct answer from each drop - down menu.
the function t relates the age of a plant fossil, in years, to the percentage, c, of carbon - 14 remaining in the fossil relative to when it was alive.
$t(c)=-3,970.9(ln c)$
use function t to complete the statements.
a fossil with 47% of its carbon - 14 remaining is approximately years old.
a fossil that is 7,000 years old will have approximately of its carbon - 14 remaining.
reset next
First Statement: A fossil with 47% of its carbon - 14 remaining is approximately how many years old?
Step 1: Identify the value of c
We know that \( c = 47\%=0.47 \). We need to substitute this value into the function \( t(c)=- 3970.9(\ln c) \).
Step 2: Calculate the natural logarithm of c
First, calculate \( \ln(0.47) \). Using a calculator, \( \ln(0.47)\approx - 0.755 \).
Step 3: Substitute into the function
Now substitute \( \ln(0.47)\approx - 0.755 \) into the function \( t(c) \):
\( t(0.47)=-3970.9\times(- 0.755) \)
\( t(0.47)\approx3970.9\times0.755 \)
\( 3970.9\times0.755 = 3970.9\times(0.7 + 0.05+0.005)=3970.9\times0.7+3970.9\times0.05 + 3970.9\times0.005=2779.63+198.545 + 19.8545 = 2997.9995\approx3000 \) (rounded to a reasonable approximation)
Second Statement: A fossil that is 7,000 years old will have approximately what percentage of its carbon - 14 remaining?
Step 1: Set up the equation
We know that \( t(c) = 7000 \) and \( t(c)=-3970.9(\ln c) \). So we set up the equation:
\( 7000=-3970.9(\ln c) \)
Step 2: Solve for \( \ln c \)
Divide both sides of the equation by \( - 3970.9 \):
\( \ln c=\frac{7000}{-3970.9}\approx - 1.763 \)
Step 3: Solve for c
To solve for \( c \), we use the exponential function (since \( y = \ln x\) and \( x = e^{y}\) are inverse functions). So \( c = e^{-1.763} \)
Using a calculator, \( e^{-1.763}\approx0.172 = 17.2\% \) (rounded to a reasonable approximation)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A fossil with 47% of its carbon - 14 remaining is approximately \(\boldsymbol{3000}\) years old.
A fossil that is 7,000 years old will have approximately \(\boldsymbol{17\%}\) (or 17.2% for more precision) of its carbon - 14 remaining.