QUESTION IMAGE
Question
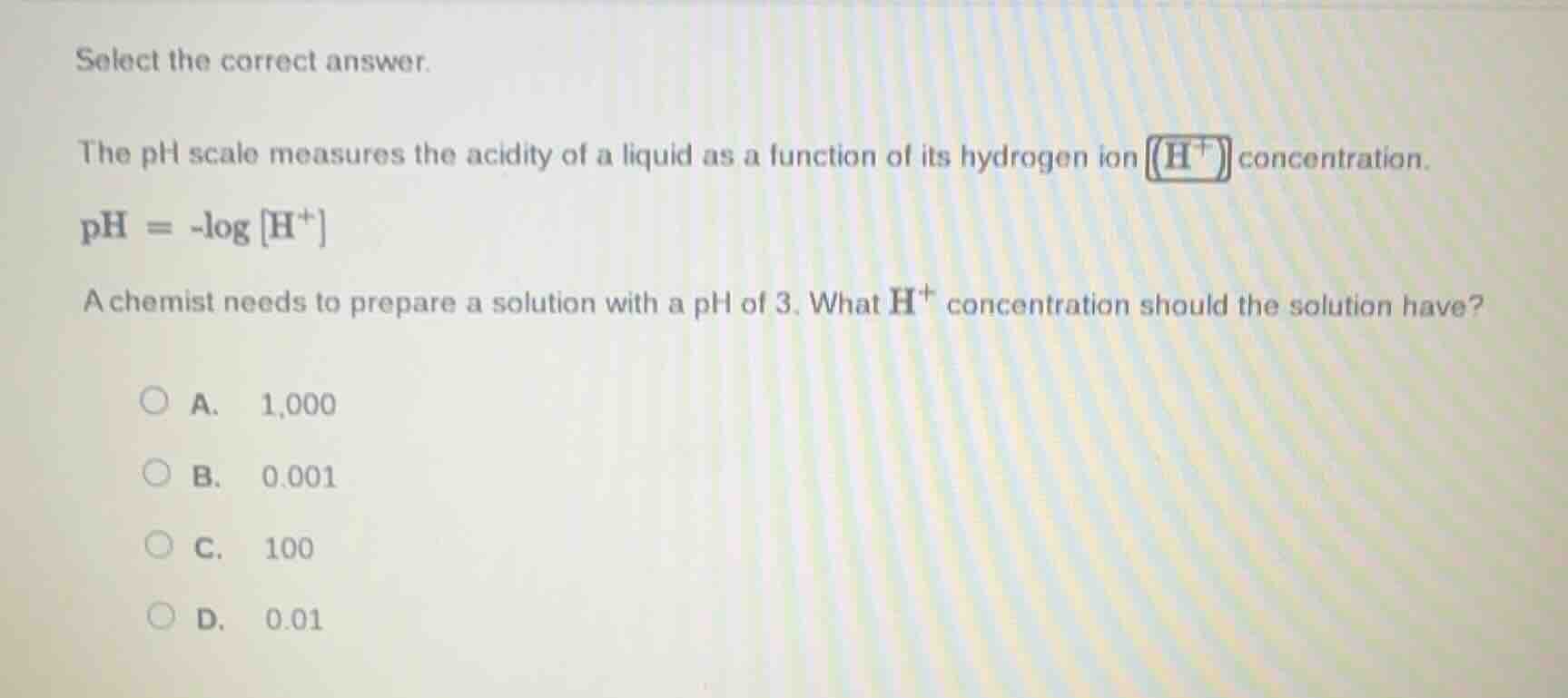
select the correct answer.
the ph scale measures the acidity of a liquid as a function of its hydrogen ion concentration.
$\text{ph} = -\log \text{h}^+$
a chemist needs to prepare a solution with a ph of 3. what $\text{h}^+$ concentration should the solution have?
a. 1,000
b. 0.001
c. 100
d. 0.01
Step1: Substitute pH into formula
Given $\text{pH} = 3$, substitute into $\text{pH} = -\log[\text{H}^+]$:
$3 = -\log[\text{H}^+]$
Step2: Rearrange to isolate log term
Multiply both sides by -1:
$\log[\text{H}^+] = -3$
Step3: Convert log to exponential form
Since $\log(x)=y$ means $10^y=x$:
$[\text{H}^+] = 10^{-3}$
Step4: Calculate the value
$10^{-3} = \frac{1}{10^3} = 0.001$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. 0.001