QUESTION IMAGE
Question
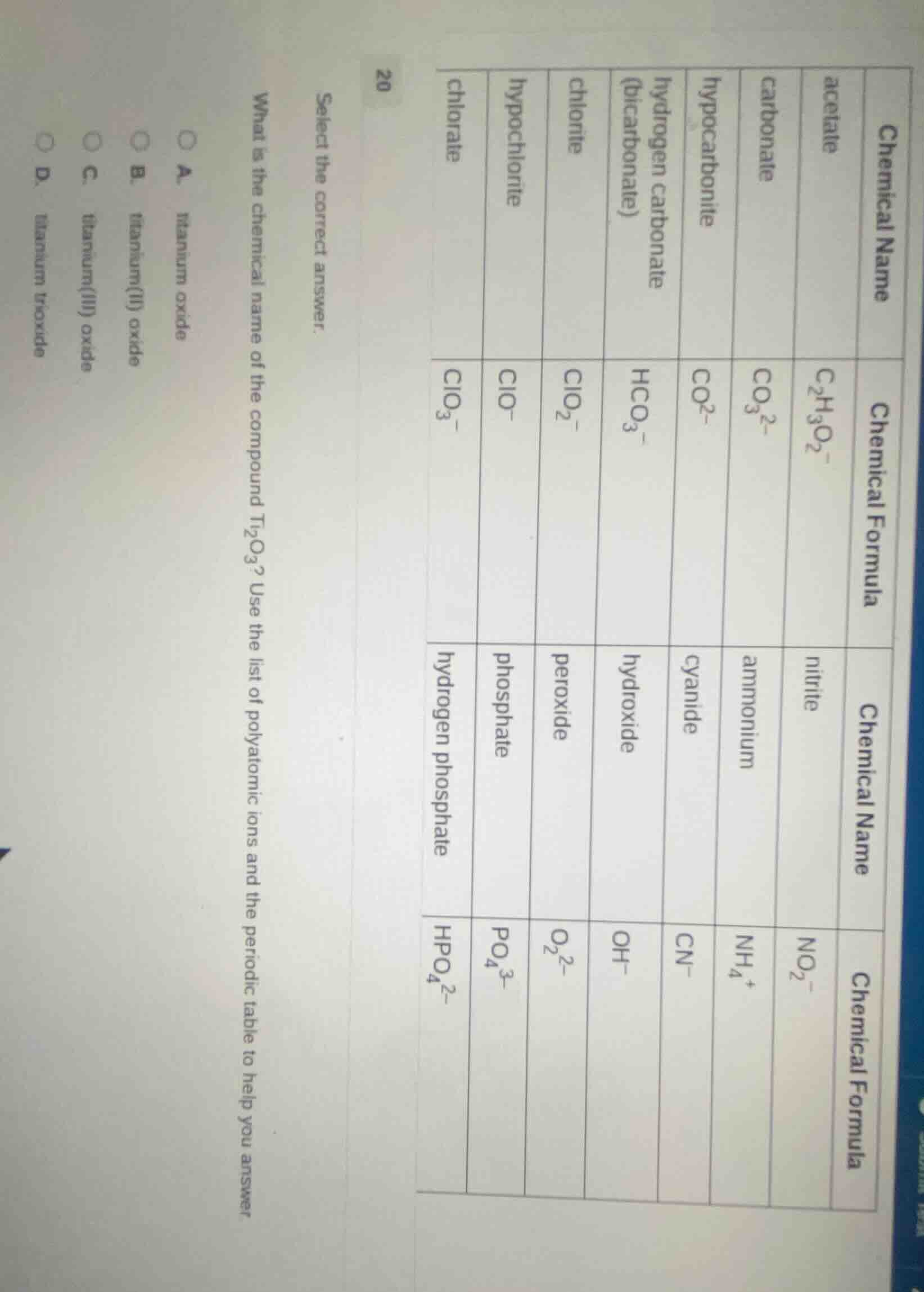
select the correct answer. what is the chemical name of the compound ti₂o₃? use the list of polyatomic ions and the periodic table to help you answer a. titanium oxide b. titanium(ii) oxide c. titanium(iii) oxide d. titanium trioxide
Step1: Determine the oxidation state of titanium
In the compound \( \text{Ti}_2\text{O}_3 \), oxygen usually has an oxidation state of \(-2\). Let the oxidation state of titanium be \( x \). Using the rule that the sum of oxidation states in a neutral compound is zero:
\( 2x + 3(-2) = 0 \)
\( 2x - 6 = 0 \)
\( 2x = 6 \)
\( x = +3 \).
Step2: Name the compound
The compound is a metal oxide (titanium and oxygen). For metals with variable oxidation states (like titanium), we use Roman numerals to indicate the oxidation state. The oxidation state of Ti is \( +3 \), so we name it as titanium(III) oxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. titanium(III) oxide