QUESTION IMAGE
Question
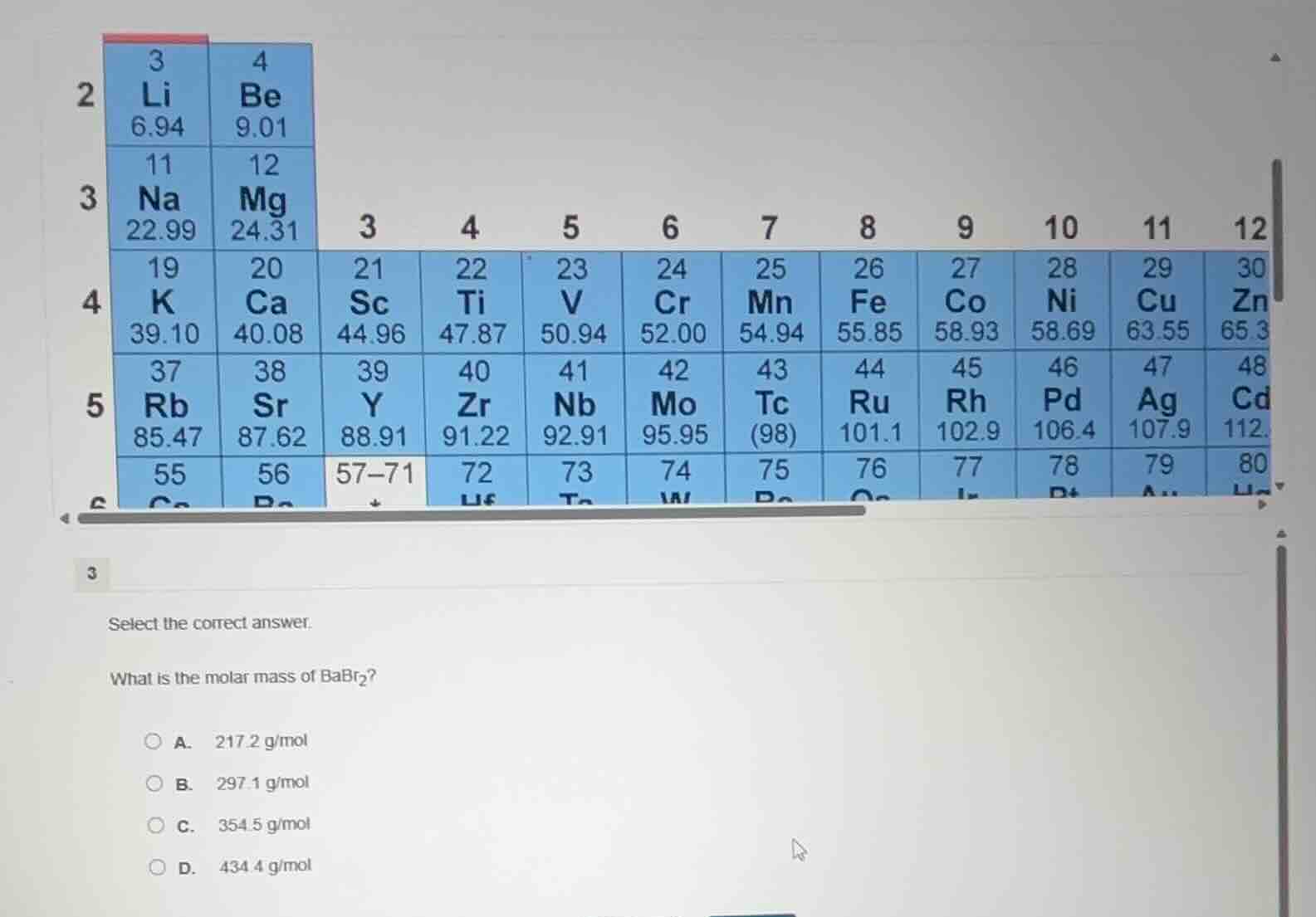
select the correct answer.
what is the molar mass of babr₂?
a. 217.2 g/mol
b. 297.1 g/mol
c. 354.5 g/mol
d. 434.4 g/mol
Step1: Find molar mass of Ba
From the periodic table, molar mass of Ba = $137.3\ \text{g/mol}$ (standard value, as Ba is element 56)
Step2: Find molar mass of Br
From standard data, molar mass of Br = $79.90\ \text{g/mol}$
Step3: Calculate total molar mass
Molar mass of $\text{BaBr}_2$ = Molar mass of Ba + 2×Molar mass of Br
$\text{Molar mass} = 137.3 + 2\times79.90$
$\text{Molar mass} = 137.3 + 159.8 = 297.1\ \text{g/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. 297.1 g/mol