QUESTION IMAGE
Question
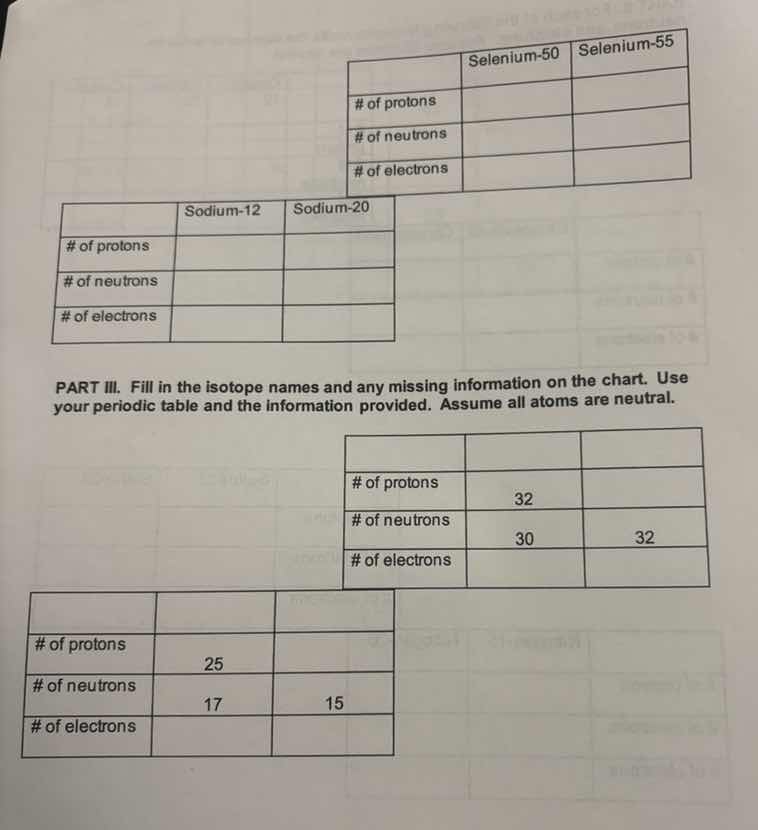
selenium-50 selenium-55
of protons
of neutrons
of electrons
sodium-12 sodium-20
of protons
of neutrons
of electrons
part iii. fill in the isotope names and any missing information on the chart. use your periodic table and the information provided. assume all atoms are neutral.
of protons 32
of neutrons 30 32
of electrons
of protons 25
of neutrons 17 15
of electrons
Step1: Find Sodium's proton count
From periodic table, sodium (Na) has atomic number 11, so protons = 11. For neutral atoms, electrons = protons = 11. Neutrons = mass number - protons.
- Sodium-12: Neutrons = $12 - 11 = 1$
- Sodium-20: Neutrons = $20 - 11 = 9$
Step2: Find Selenium's proton count
Selenium (Se) has atomic number 34, so protons = 34. For neutral atoms, electrons = 34. Neutrons = mass number - protons.
- Selenium-50: Neutrons = $50 - 34 = 16$
- Selenium-55: Neutrons = $55 - 34 = 21$
Step3: Solve middle Se isotope table
First column: Protons=32 (germanium, Ge), neutrons=30, mass number = $32 + 30 = 62$, so isotope is Germanium-62. Electrons=32 (neutral atom).
Second column: Protons=32, neutrons=32, mass number = $32 + 32 = 64$, so isotope is Germanium-64. Electrons=32.
Step4: Solve bottom manganese table
First column: Protons=25 (manganese, Mn), neutrons=17, mass number = $25 + 17 = 42$, isotope is Manganese-42. Electrons=25 (neutral atom).
Second column: Protons=25, neutrons=15, mass number = $25 + 15 = 40$, isotope is Manganese-40. Electrons=25.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Sodium Table
| Sodium-12 | Sodium-20 | |
|---|---|---|
| # of neutrons | 1 | 9 |
| # of electrons | 11 | 11 |
Selenium Table
| Selenium-50 | Selenium-55 | |
|---|---|---|
| # of neutrons | 16 | 21 |
| # of electrons | 34 | 34 |
Middle Isotope Table
| Germanium-62 | Germanium-64 | |
|---|---|---|
| # of neutrons | 30 | 32 |
| # of electrons | 32 | 32 |
Bottom Isotope Table
| Manganese-42 | Manganese-40 | |
|---|---|---|
| # of neutrons | 17 | 15 |
| # of electrons | 25 | 25 |