QUESTION IMAGE
Question
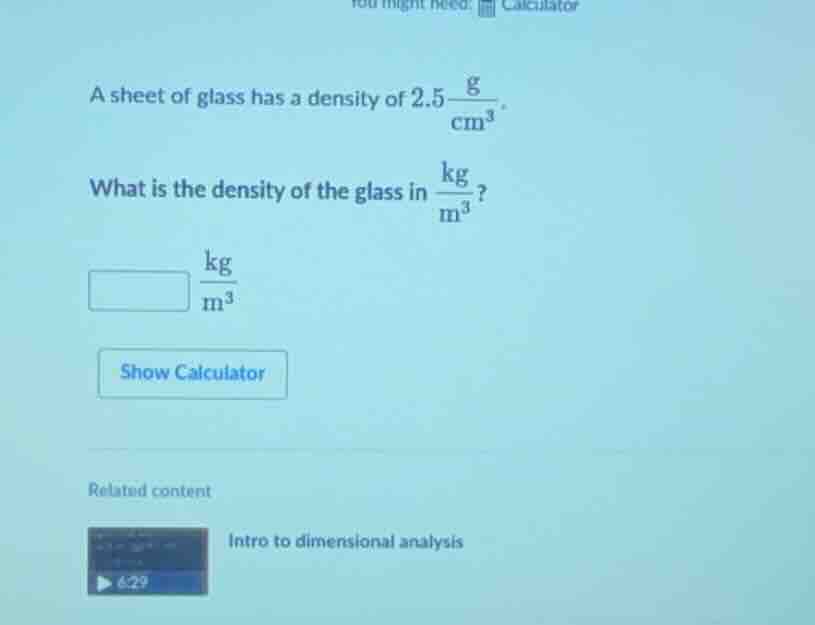
a sheet of glass has a density of $2.5\frac{\text{g}}{\text{cm}^3}$. what is the density of the glass in $\frac{\text{kg}}{\text{m}^3}$? \boxed{} $\frac{\text{kg}}{\text{m}^3}$ show calculator related content intro to dimensional analysis 6:29
Step1: Convert grams to kilograms
We know that \(1\space kg = 1000\space g\), so to convert grams to kilograms, we divide by \(1000\). So \(2.5\space g\) is equal to \(2.5\div1000 = 2.5\times10^{- 3}\space kg\).
Step2: Convert cubic centimeters to cubic meters
We know that \(1\space m=100\space cm\), so \(1\space m^{3}=(100\space cm)^{3}=100^{3}\space cm^{3} = 10^{6}\space cm^{3}\). So to convert \(cm^{3}\) to \(m^{3}\), we divide by \(10^{6}\). So \(\frac{1}{cm^{3}}=\frac{10^{6}}{m^{3}}\).
Step3: Calculate the density in \(\frac{kg}{m^{3}}\)
The density is \(2.5\space\frac{g}{cm^{3}}\). Substituting the conversions from step 1 and step 2, we get:
\(2.5\space\frac{g}{cm^{3}}=2.5\times\frac{10^{-3}\space kg}{10^{- 6}\space m^{3}}\)
\(=2.5\times10^{-3 + 6}\space\frac{kg}{m^{3}}\)
\(=2.5\times10^{3}\space\frac{kg}{m^{3}}\)
\( = 2500\space\frac{kg}{m^{3}}\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(2500\)