QUESTION IMAGE
Question
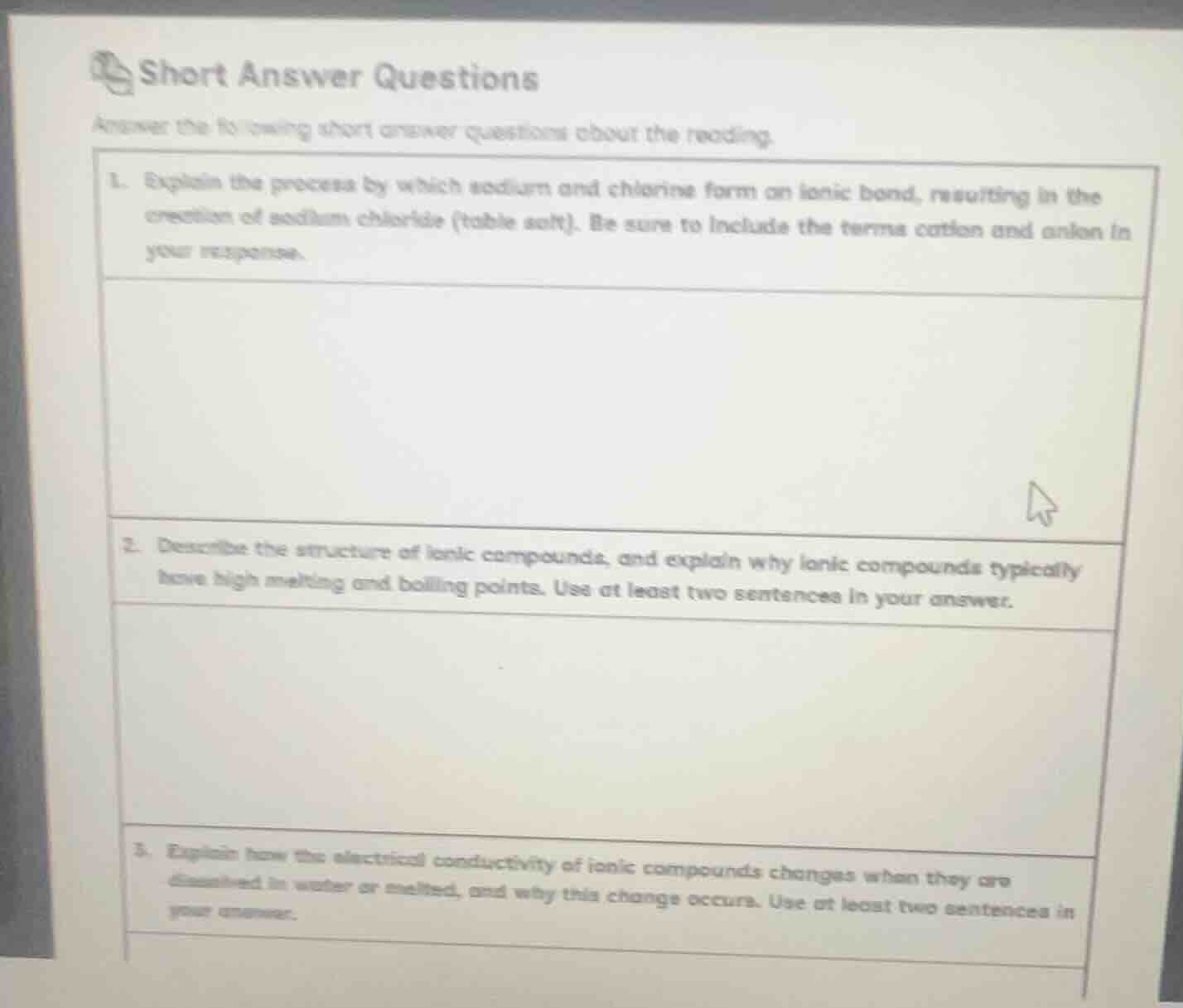
short answer questions
answer the following short answer questions about the reading.
- explain the process by which sodium and chlorine form an ionic bond, resulting in the creation of sodium chloride (table salt). be sure to include the terms cation and anion in your response.
- describe the structure of ionic compounds, and explain why ionic compounds typically have high melting and boiling points. use at least two sentences in your answer.
- explain how the electrical conductivity of ionic compounds changes when they are dissolved in water or melted, and why this change occurs. use at least two sentences in your answer.
Brief Explanations
- For sodium and chlorine ionic bonding: Sodium (a group 1 metal) has 1 valence electron, which it loses to form a positively charged cation ($\text{Na}^+$). Chlorine (a group 17 nonmetal) has 7 valence electrons, so it gains this electron to form a negatively charged anion ($\text{Cl}^-$). The electrostatic attraction between the oppositely charged $\text{Na}^+$ and $\text{Cl}^-$ creates the ionic bond in sodium chloride.
- For ionic compound structure and melting/boiling points: Ionic compounds form a crystalline lattice structure, where positively charged cations and negatively charged anions are arranged in a repeating, tightly packed 3D pattern. This lattice is held together by strong electrostatic attractions between the oppositely charged ions; a large amount of thermal energy is required to break these strong interionic forces, which is why ionic compounds have high melting and boiling points.
- For electrical conductivity changes: In their solid state, ionic compounds do not conduct electricity because their ions are locked in a rigid lattice and cannot move freely. When melted or dissolved in water, the lattice breaks apart, freeing the cations and anions to move independently; these mobile charged particles are able to carry an electric current, so the compound becomes electrically conductive.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Sodium loses its single valence electron to become a positively charged cation ($\text{Na}^+$), while chlorine gains this electron to become a negatively charged anion ($\text{Cl}^-$). The electrostatic attraction between these oppositely charged ions forms the ionic bond that creates sodium chloride.
- Ionic compounds have a repeating 3D crystalline lattice structure made of tightly packed, oppositely charged cations and anions. Strong electrostatic forces hold this lattice together, so a large amount of energy is needed to break these interactions, resulting in high melting and boiling points.
- Solid ionic compounds do not conduct electricity because their ions are trapped in a fixed lattice and cannot move. When melted or dissolved in water, the lattice breaks, releasing mobile, charged ions that can carry an electric current, making the compound conductive.