QUESTION IMAGE
Question
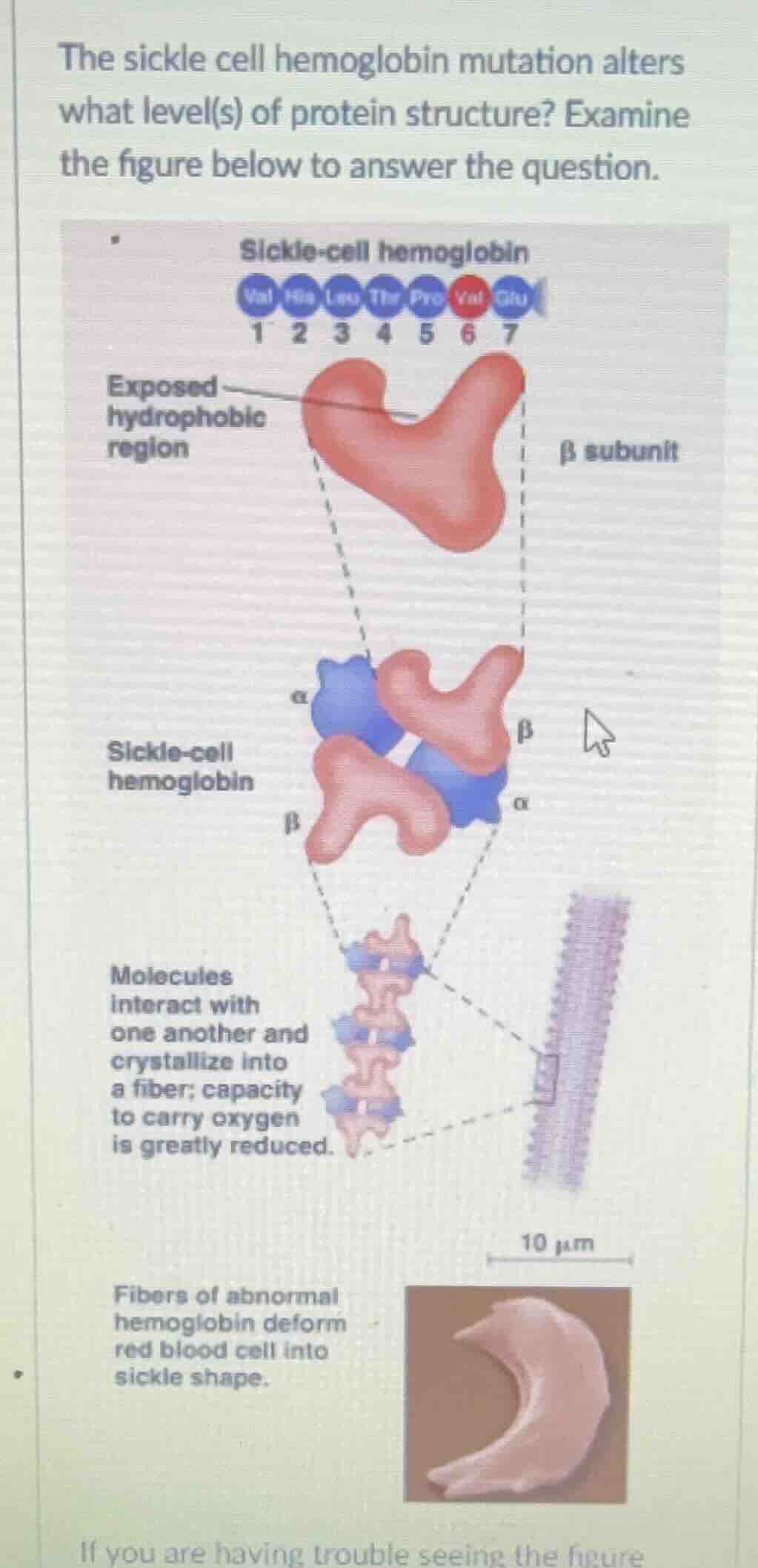
the sickle cell hemoglobin mutation alters what level(s) of protein structure? examine the figure below to answer the question.
Brief Explanations
- Primary (1°) Structure: The sickle - cell mutation changes the amino acid sequence of the hemoglobin's β - subunit (e.g., a glutamic acid (Glu) is replaced by a valine (Val) at position 6). The primary structure of a protein is defined by its linear sequence of amino acids.
- Tertiary (3°) Structure: The substitution of a hydrophilic amino acid (Glu) with a hydrophobic one (Val) leads to the exposure of a hydrophobic region on the β - subunit. This affects the folding of the polypeptide chain, as hydrophobic interactions are important for maintaining the three - dimensional (tertiary) structure of the protein. The tertiary structure is the overall three - dimensional shape of a single polypeptide chain, which is determined by interactions like hydrophobic interactions, hydrogen bonds, ionic bonds, and disulfide bridges.
- Quaternary (4°) Structure: The abnormal tertiary structure of the β - subunits causes the sickle - cell hemoglobin molecules to interact with each other. Hemoglobin is a tetramer (has four subunits: 2 α and 2 β). The abnormal interaction between the β - subunits (due to the exposed hydrophobic regions) leads to the formation of long fibers of hemoglobin. The quaternary structure of a protein refers to the way multiple polypeptide chains (subunits) are arranged and interact with each other to form a functional protein complex. The formation of these fibers disrupts the normal quaternary structure of hemoglobin, which in turn affects the shape and function of red blood cells.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The sickle - cell hemoglobin mutation alters the primary, tertiary, and quaternary levels of protein structure.