QUESTION IMAGE
Question
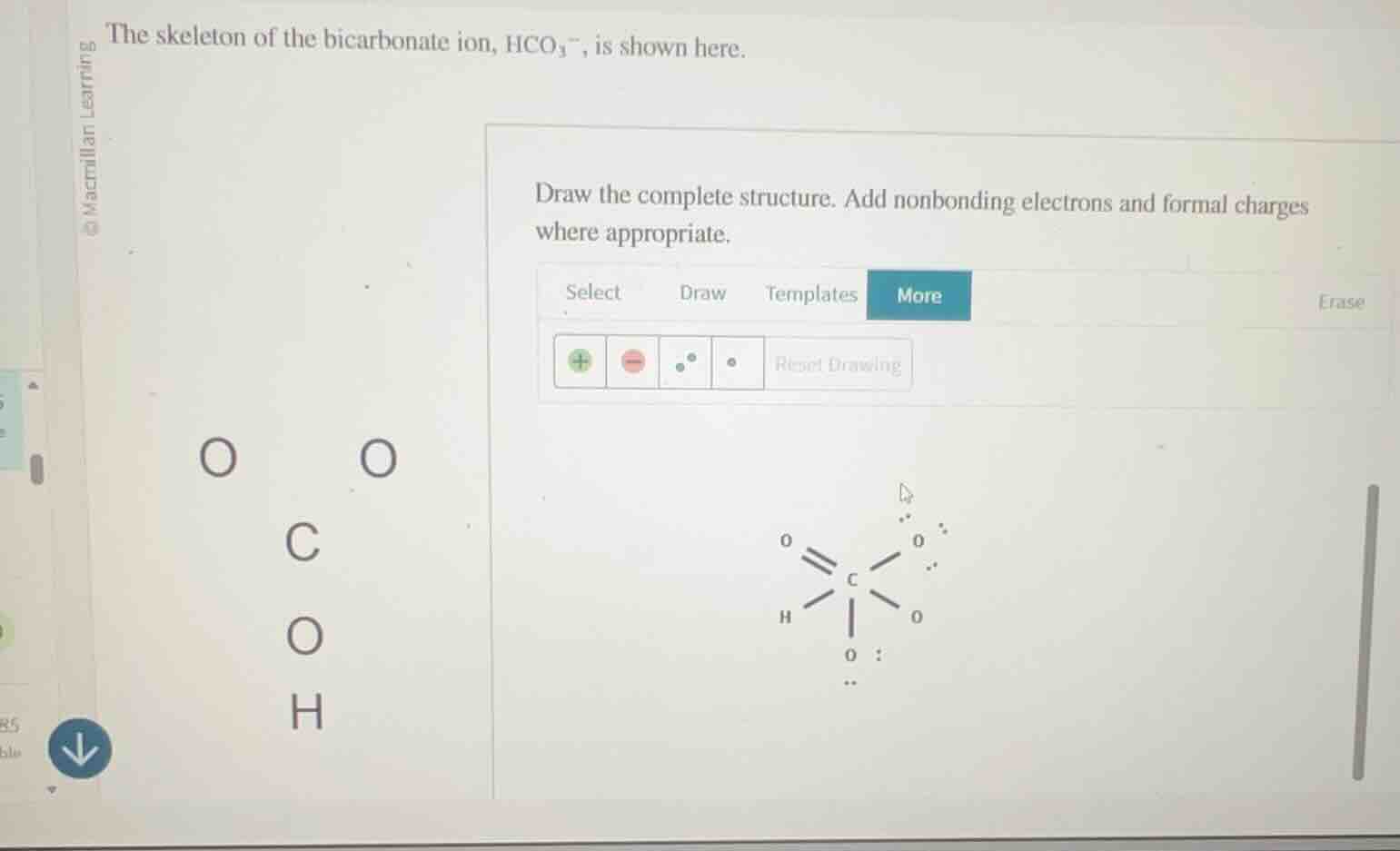
the skeleton of the bicarbonate ion, hco₃⁻, is shown here. draw the complete structure. add nonbonding electrons and formal charges where appropriate.
Step1: Calculate valence electrons
Total valence electrons: $4(\text{C}) + 3\times6(\text{O}) + 1(\text{H}) + 1(\text{charge}) = 4+18+1+1=24$
Step2: Assign bonding electrons
Skeleton has 4 bonds (C=O, C-O, C-OH, C-H), using $4\times2=8$ electrons. Remaining electrons: $24-8=16$
Step3: Add lone pairs to O atoms
- Double-bonded O: 2 lone pairs ($4$ electrons)
- Single-bonded O (not bonded to H): 3 lone pairs ($6$ electrons), formal charge: $6 - (6 + 1) = -1$
- O in OH: 2 lone pairs ($4$ electrons), H bonded to this O
- Remaining 2 electrons: none, total used: $8+4+6+4+2=24$
Step4: Verify formal charges
C: $4 - (4 + 0) = 0$; H: $1 - (1 + 0) = 0$; Double-bonded O: $6 - (4 + 2) = 0$; OH O: $6 - (4 + 2) = 0$; Single-bonded O: $6 - (6 + 1) = -1$; Total charge: $0+0+0+0-1=-1$ (matches ion charge)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The correct Lewis structure is:
..
O = C - O⁻
| ..
O-H
..(Or with spatial arrangement: Carbon is central, double-bonded to one O (with 2 lone pairs), single-bonded to one O (with 3 lone pairs and a -1 formal charge), single-bonded to an -OH group (the O has 2 lone pairs, bonded to H). All electrons and charges balance to match the $\text{HCO}_3^-$ ion.)