QUESTION IMAGE
Question
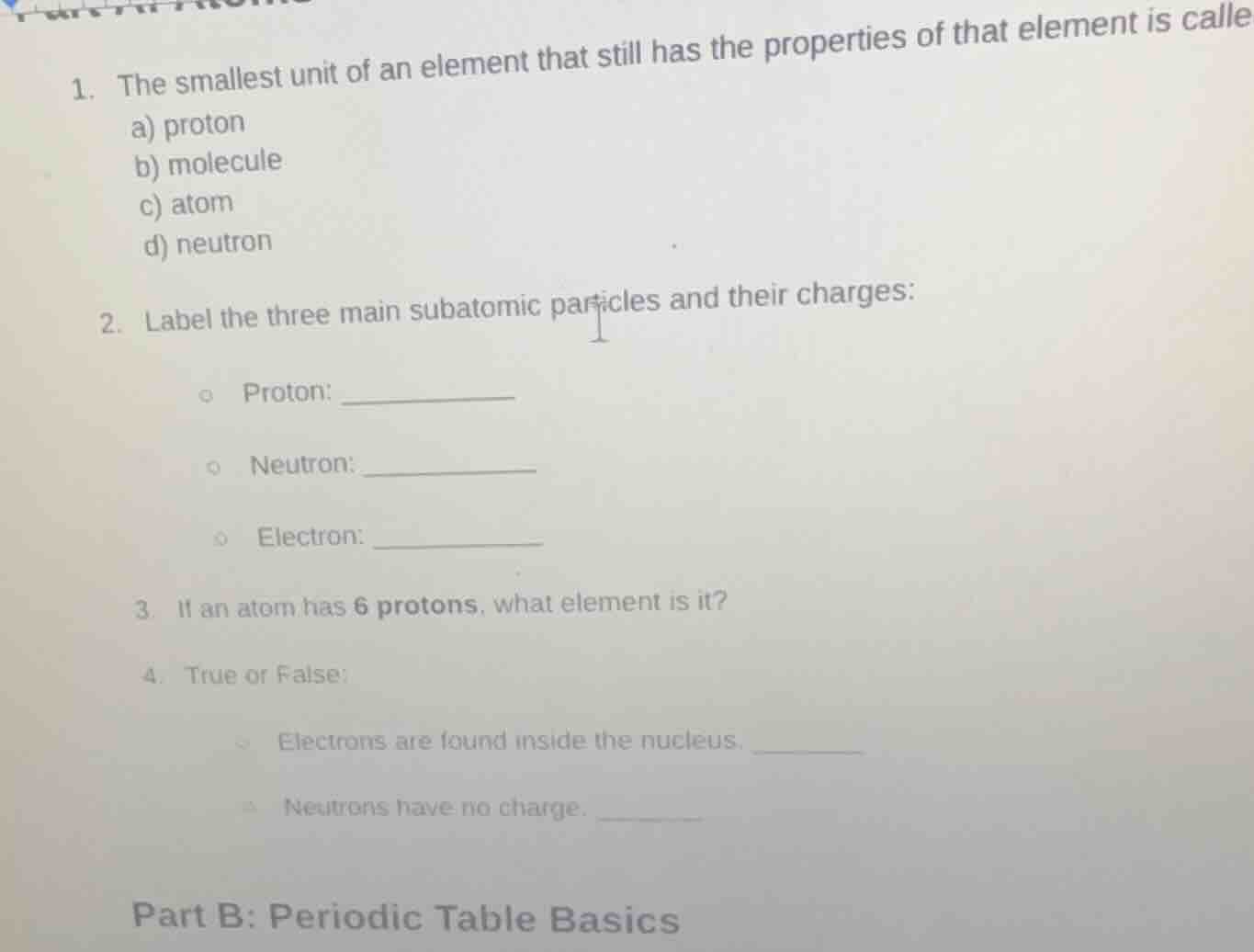
- the smallest unit of an element that still has the properties of that element is calle
a) proton
b) molecule
c) atom
d) neutron
- label the three main subatomic particles and their charges:
- proton: _________
- neutron: _________
- electron: _________
- if an atom has 6 protons, what element is it?
- true or false:
- electrons are found inside the nucleus. ______
- neutrons have no charge. ______
part b: periodic table basics
Response
Question 1
Brief Explanations
A proton is a subatomic particle, a molecule is made of multiple atoms, and a neutron is also a subatomic particle. An atom is the smallest unit of an element that retains the element's properties.
Brief Explanations
Protons are positively charged, neutrons have no charge (neutral), and electrons are negatively charged. This is based on atomic structure knowledge.
Brief Explanations
The number of protons in an atom is its atomic number. Carbon has an atomic number of 6, meaning it has 6 protons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
c) atom