QUESTION IMAGE
Question
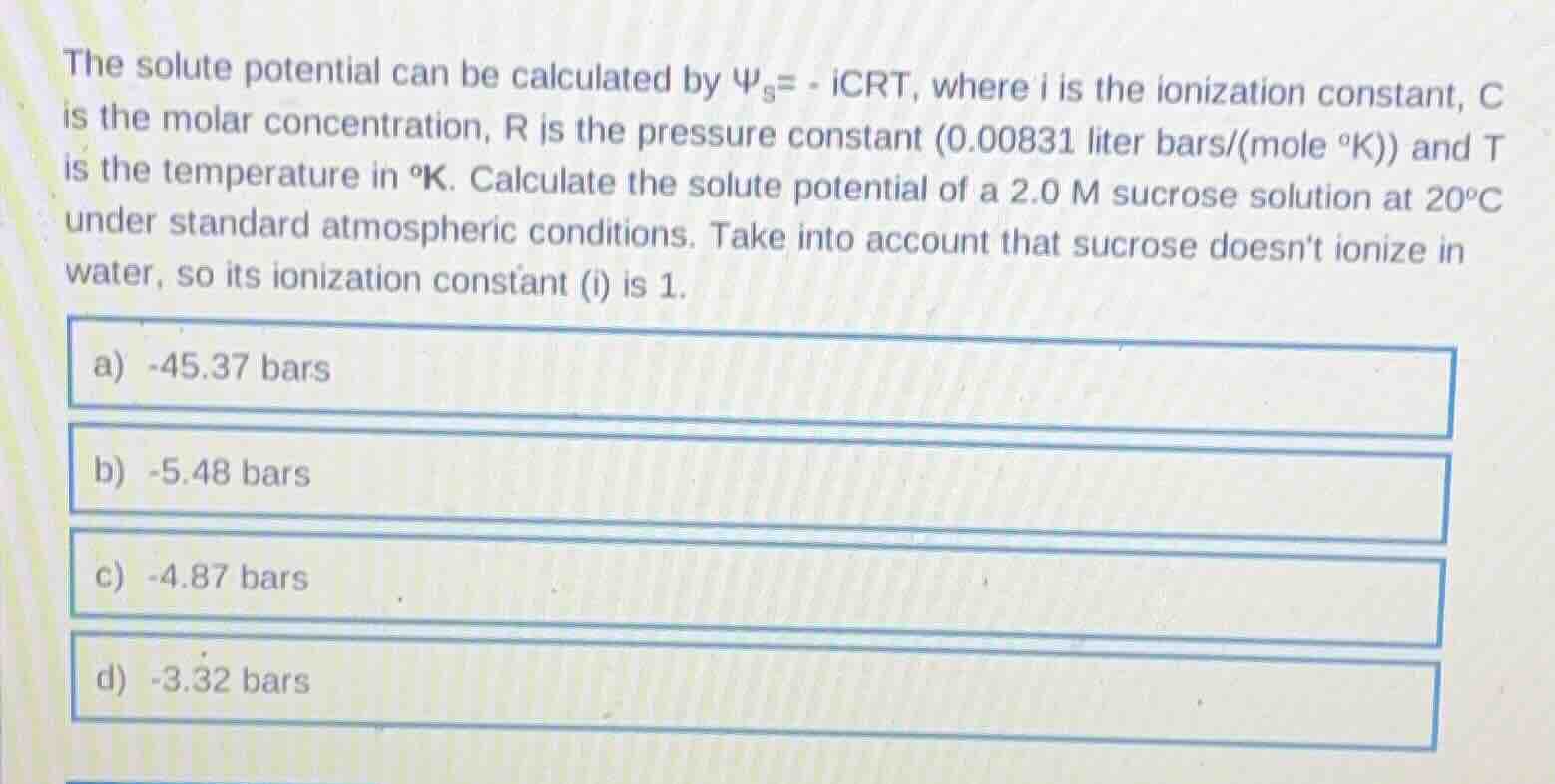
the solute potential can be calculated by $\psi_s = -icrt$, where $i$ is the ionization constant, $c$ is the molar concentration, $r$ is the pressure constant (0.00831 liter bars/(mole $^\circ$k)) and $t$ is the temperature in $^\circ$k. calculate the solute potential of a 2.0 m sucrose solution at 20$^\circ$c under standard atmospheric conditions. take into account that sucrose doesnt ionize in water, so its ionization constant ($i$) is 1.\
a) -45.37 bars\
b) -5.48 bars\
c) -4.87 bars\
d) -3.32 bars
Step1: Convert temp to Kelvin
$T = 20 + 273 = 293 \ ^\circ\text{K}$
Step2: List known values
$i=1$, $C=2.0\ \text{M}$, $R=0.00831\ \text{liter bars/(mole }^\circ\text{K)}$, $T=293\ ^\circ\text{K}$
Step3: Substitute into formula
$\Psi_s = -iCRT = -1 \times 2.0 \times 0.00831 \times 293$
Step4: Calculate the value
$\Psi_s = -2.0 \times 0.00831 \times 293 = -48.7 \times 0.00831 \times 10 = -4.87$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) -45.37 bars
b) -5.48 bars
c) -4.87 bars
d) -3.32 bars
The correct answer is c) -4.87 bars