QUESTION IMAGE
Question
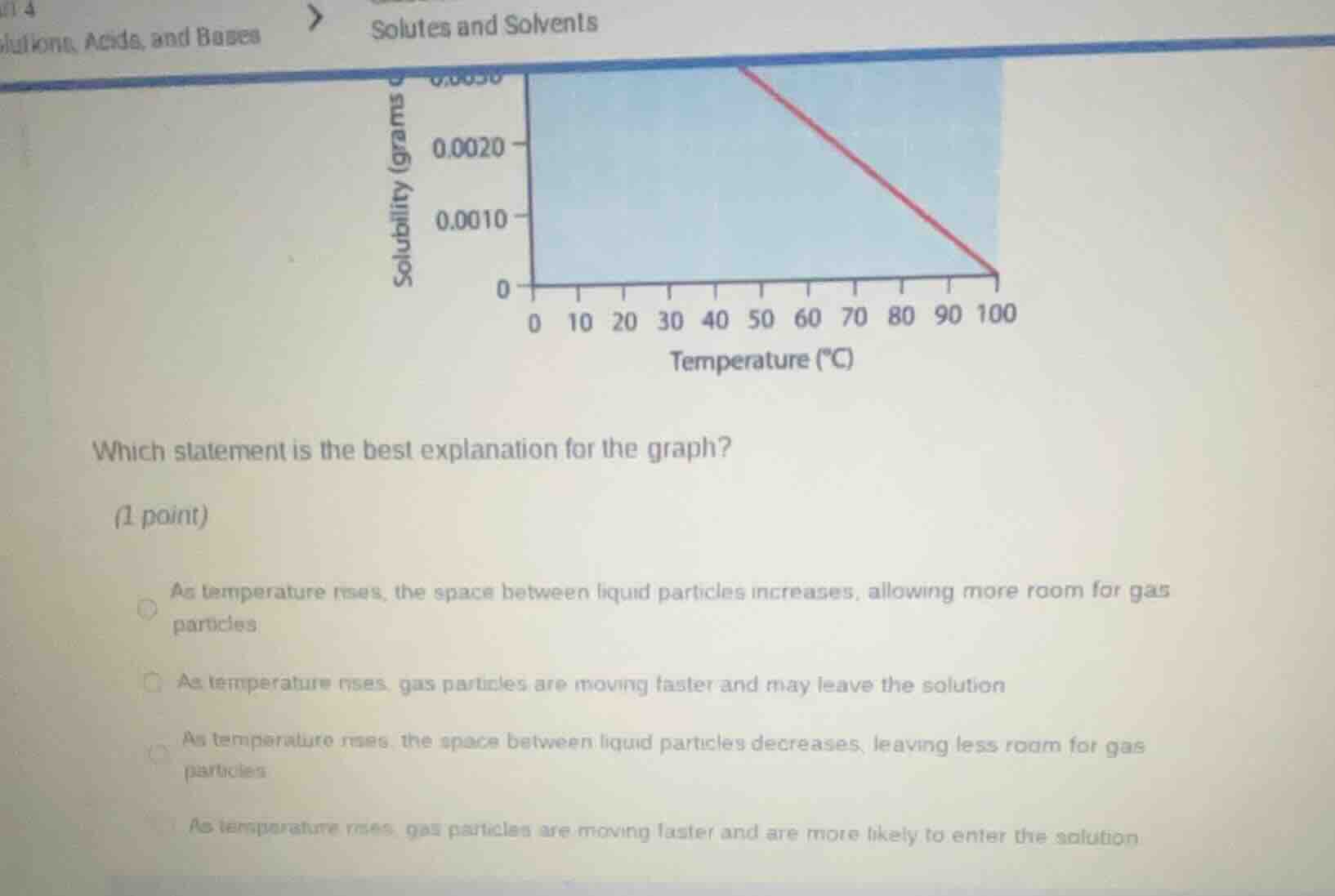
solutes and solvents
which statement is the best explanation for the graph?
(1 point)
as temperature rises, the space between liquid particles increases, allowing more room for gas particles
as temperature rises, gas particles are moving faster and may leave the solution
as temperature rises, the space between liquid particles decreases, leaving less room for gas particles
as temperature rises, gas particles are moving faster and are more likely to enter the solution
The graph shows that solubility of a substance decreases as temperature increases, which is a property of gaseous solutes in liquids. When temperature rises, gas particles gain kinetic energy, move faster, and have enough energy to escape the liquid solution, reducing their solubility.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. As temperature rises, gas particles are moving faster and may leave the solution