QUESTION IMAGE
Question
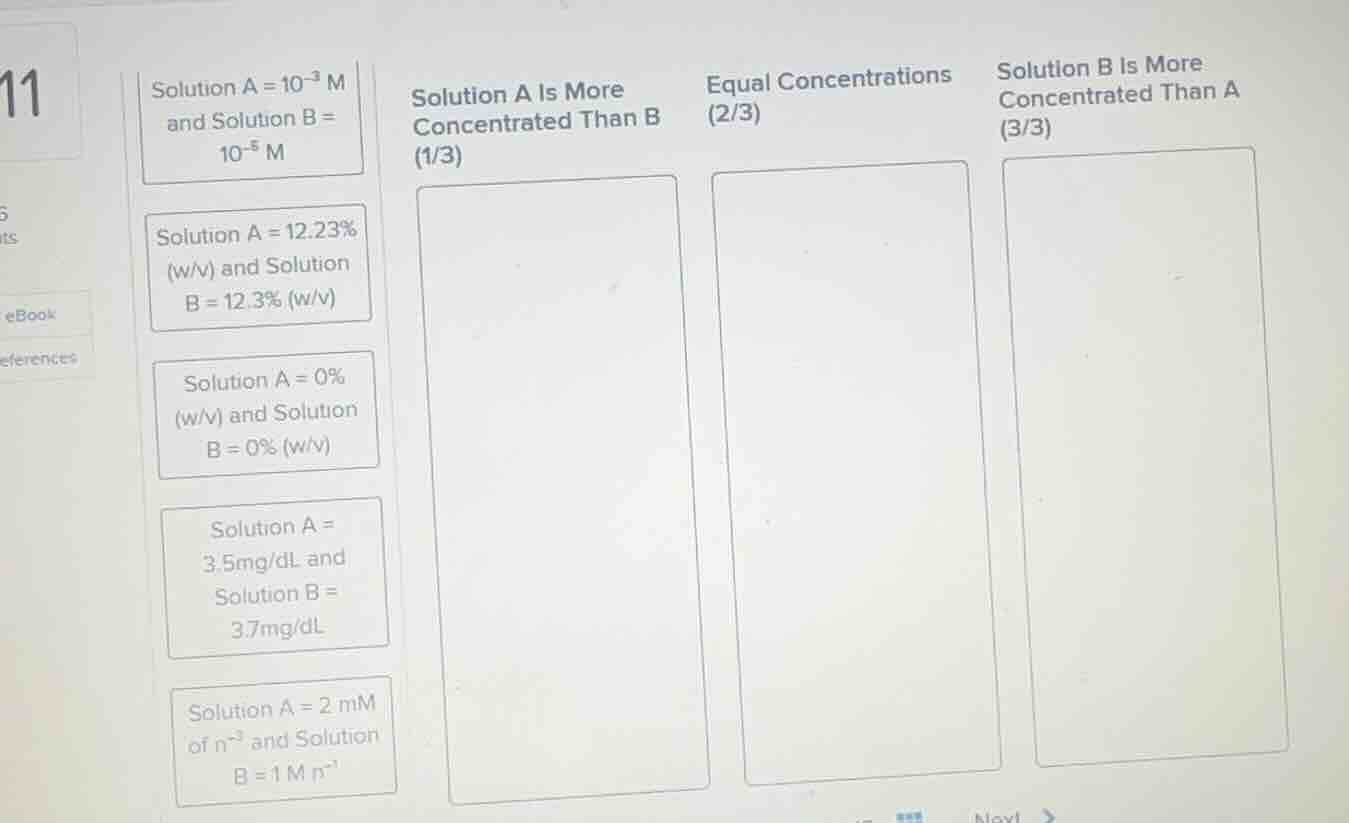
solution a = 10⁻³ m and solution b = 10⁻⁵ m
solution a = 12.23% (w/v) and solution b = 12.3% (w/v)
solution a = 0% (w/v) and solution b = 0% (w/v)
solution a = 3.5mg/dl and solution b = 3.7mg/dl
solution a = 2 mm of n⁻³ and solution b = 1 m n⁻¹
solution a is more concentrated than b (1/3)
equal concentrations (2/3)
solution b is more concentrated than a (3/3)
Step1: Compare molar concentrations
$10^{-3}\ \text{M} > 10^{-5}\ \text{M}$, so Solution A is more concentrated.
Step2: Compare (w/v) percentages
$12.23\% < 12.3\%$, so Solution B is more concentrated.
Step3: Compare 0% (w/v) solutions
$0\% = 0\%$, so concentrations are equal.
Step4: Compare mg/dL concentrations
$3.5\ \text{mg/dL} < 3.7\ \text{mg/dL}$, so Solution B is more concentrated.
Step5: Compare molar ion concentrations
$2\ \text{mM }n^{2+} = 2\times10^{-3}\ \text{M }n^{2+}$; $1\ \text{M }n^- = 1\ \text{M }n^-$. For equal cation/anion equivalence, $2\times10^{-3}\ \text{M }n^{2+}$ has equivalent concentration to $4\times10^{-3}\ \text{M }n^-$, which is less than $1\ \text{M }n^-$, so Solution B is more concentrated.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Solution A Is More Concentrated Than B:
- Solution A = $10^{-3}$ M and Solution B = $10^{-5}$ M
Equal Concentrations:
- Solution A = 0% (w/v) and Solution B = 0% (w/v)
Solution B Is More Concentrated Than A:
- Solution A = 12.23% (w/v) and Solution B = 12.3% (w/v)
- Solution A = 3.5mg/dL and Solution B = 3.7mg/dL
- Solution A = 2 mM of $n^{2+}$ and Solution B = 1 M $n^-$