QUESTION IMAGE
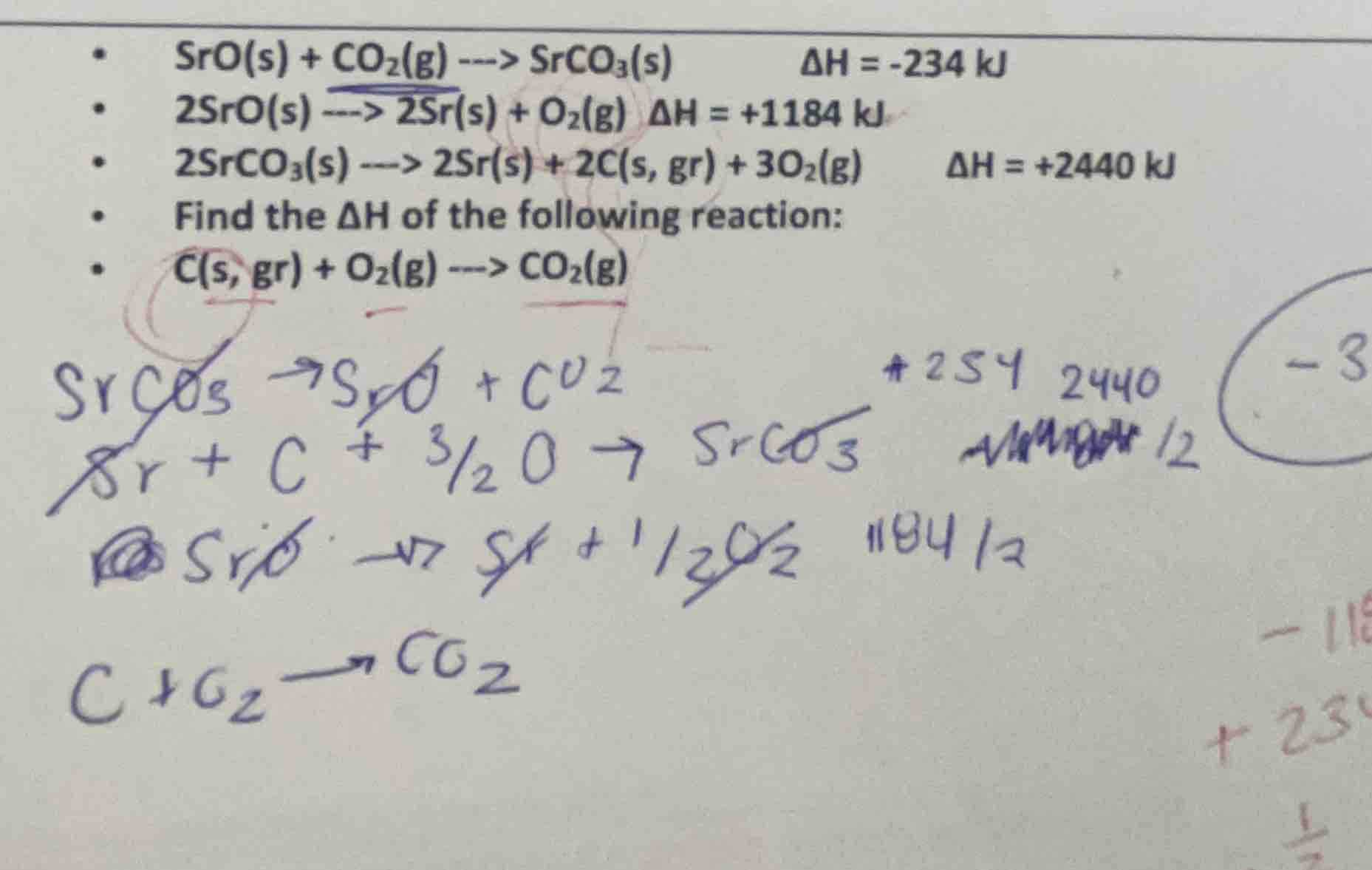
Question
- sro(s) + co₂(g) ---> srco₃(s) δh = -234 kj• 2sro(s) ---> 2sr(s) + o₂(g) δh = +1184 kj• 2srco₃(s) ---> 2sr(s) + 2c(s, gr) + 3o₂(g) δh = +2440 kj• find the δh of the following reaction:• c(s, gr) + o₂(g) ---> co₂(g)
Step1: Label the given reactions
Let's label the given reactions as follows:
Reaction 1: $\ce{SrO(s) + CO_{2}(g) -> SrCO_{3}(s)}$ $\Delta H_1 = -234\ \text{kJ}$
Reaction 2: $\ce{2SrO(s) -> 2Sr(s) + O_{2}(g)}$ $\Delta H_2 = +1184\ \text{kJ}$
Reaction 3: $\ce{2SrCO_{3}(s) -> 2Sr(s) + 2C(s, gr) + 3O_{2}(g)}$ $\Delta H_3 = +2440\ \text{kJ}$
Target reaction: $\ce{C(s, gr) + O_{2}(g) -> CO_{2}(g)}$
Step2: Manipulate Reaction 1
Reverse Reaction 1 and multiply by 2:
$2\ce{SrCO_{3}(s) -> 2SrO(s) + 2CO_{2}(g)}$ $\Delta H_{1}' = -2\times(-234) = +468\ \text{kJ}$ (Reversed, so sign flips; multiplied by 2, so $\Delta H$ multiplies by 2)
Step3: Manipulate Reaction 2
Divide Reaction 2 by 2:
$\ce{SrO(s) -> Sr(s) + \frac{1}{2}O_{2}(g)}$ $\Delta H_{2}' = \frac{1184}{2} = +592\ \text{kJ}$ (Divided by 2, so $\Delta H$ divides by 2)
Step4: Manipulate Reaction 3
Divide Reaction 3 by 2:
$\ce{SrCO_{3}(s) -> Sr(s) + C(s, gr) + \frac{3}{2}O_{2}(g)}$ $\Delta H_{3}' = \frac{2440}{2} = +1220\ \text{kJ}$ (Divided by 2, so $\Delta H$ divides by 2)
Step5: Combine the manipulated reactions
Now, let's write the manipulated reactions:
- $2\ce{SrCO_{3}(s) -> 2SrO(s) + 2CO_{2}(g)}$ $\Delta H_{1}' = +468\ \text{kJ}$
- $2\ce{SrO(s) -> 2Sr(s) + O_{2}(g)}$ (Wait, no, we divided Reaction 2 by 2 earlier. Let's correct. Let's use the divided Reaction 2: $\ce{SrO(s) -> Sr(s) + \frac{1}{2}O_{2}(g)}$ $\Delta H_{2}' = +592\ \text{kJ}$. But we have 2 moles in Reaction 1's product (2SrO), so multiply this by 2: $2\ce{SrO(s) -> 2Sr(s) + O_{2}(g)}$ $\Delta H_{2}'' = 2\times592 = +1184\ \text{kJ}$ (Wait, no, original Reaction 2 is $2SrO -> 2Sr + O2$, so maybe better to use Reaction 3 and the reversed Reaction 1 and Reaction 2.
Alternative approach: Let's express the target reaction in terms of the given reactions.
Target reaction: $\ce{C + O2 -> CO2}$
From Reaction 3: $\ce{SrCO3 -> Sr + C + 3/2 O2}$ $\Delta H_3' = +1220\ \text{kJ}$ (divided by 2)
From Reaction 1 reversed: $\ce{SrCO3 -> SrO + CO2}$ $\Delta H_1' = +234\ \text{kJ}$ (reversed, so $\Delta H = +234$; no multiplication yet)
From Reaction 2 divided: $\ce{SrO -> Sr + 1/2 O2}$ $\Delta H_2' = +592\ \text{kJ}$ (divided by 2)
Wait, maybe a better way: Let's use Hess's law. The target reaction is $\ce{C + O2 -> CO2}$. Let's find the sum of reactions to get this.
Let's take Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ $\Delta H3 = +2440$
Reverse Reaction 1 (times 2): $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H1' = +468$ (since original Reaction 1: $\ce{SrO + CO2 -> SrCO3}$ $\Delta H = -234$, so reversed is $\ce{SrCO3 -> SrO + CO2}$ $\Delta H = +234$, times 2: $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H = +468$)
Reverse Reaction 2: $\ce{2Sr + O2 -> 2SrO}$ $\Delta H2' = -1184$ (since original Reaction 2: $\ce{2SrO -> 2Sr + O2}$ $\Delta H = +1184$, reversed is $\ce{2Sr + O2 -> 2SrO}$ $\Delta H = -1184$)
Now, add Reaction 3, reversed Reaction 1 (times 2), and reversed Reaction 2:
Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ $\Delta H3 = +2440$
Reversed Reaction 1 (times 2): $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H1' = +468$ (Wait, no, reversed Reaction 1 is $\ce{SrCO3 -> SrO + CO2}$ $\Delta H = +234$, so times 2 is $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H = +468$. But if we add Reaction 3 and reversed Reaction 1 (times 2), we get:
$\ce{2SrCO3 + 2SrCO3 -> 2Sr + 2C + 3O2 + 2SrO + 2CO2}$ No, that's not right. Let's correct.
Wait, the target reaction is $\ce{C + O2 -> CO2}$. Let's express each reaction in terms of C, O2, CO2.
From Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ $\Delta H3 = +2440$ → divide by 2: $\ce{SrCO3 -> Sr…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Step1: Label the given reactions
Let's label the given reactions as follows:
Reaction 1: $\ce{SrO(s) + CO_{2}(g) -> SrCO_{3}(s)}$ $\Delta H_1 = -234\ \text{kJ}$
Reaction 2: $\ce{2SrO(s) -> 2Sr(s) + O_{2}(g)}$ $\Delta H_2 = +1184\ \text{kJ}$
Reaction 3: $\ce{2SrCO_{3}(s) -> 2Sr(s) + 2C(s, gr) + 3O_{2}(g)}$ $\Delta H_3 = +2440\ \text{kJ}$
Target reaction: $\ce{C(s, gr) + O_{2}(g) -> CO_{2}(g)}$
Step2: Manipulate Reaction 1
Reverse Reaction 1 and multiply by 2:
$2\ce{SrCO_{3}(s) -> 2SrO(s) + 2CO_{2}(g)}$ $\Delta H_{1}' = -2\times(-234) = +468\ \text{kJ}$ (Reversed, so sign flips; multiplied by 2, so $\Delta H$ multiplies by 2)
Step3: Manipulate Reaction 2
Divide Reaction 2 by 2:
$\ce{SrO(s) -> Sr(s) + \frac{1}{2}O_{2}(g)}$ $\Delta H_{2}' = \frac{1184}{2} = +592\ \text{kJ}$ (Divided by 2, so $\Delta H$ divides by 2)
Step4: Manipulate Reaction 3
Divide Reaction 3 by 2:
$\ce{SrCO_{3}(s) -> Sr(s) + C(s, gr) + \frac{3}{2}O_{2}(g)}$ $\Delta H_{3}' = \frac{2440}{2} = +1220\ \text{kJ}$ (Divided by 2, so $\Delta H$ divides by 2)
Step5: Combine the manipulated reactions
Now, let's write the manipulated reactions:
- $2\ce{SrCO_{3}(s) -> 2SrO(s) + 2CO_{2}(g)}$ $\Delta H_{1}' = +468\ \text{kJ}$
- $2\ce{SrO(s) -> 2Sr(s) + O_{2}(g)}$ (Wait, no, we divided Reaction 2 by 2 earlier. Let's correct. Let's use the divided Reaction 2: $\ce{SrO(s) -> Sr(s) + \frac{1}{2}O_{2}(g)}$ $\Delta H_{2}' = +592\ \text{kJ}$. But we have 2 moles in Reaction 1's product (2SrO), so multiply this by 2: $2\ce{SrO(s) -> 2Sr(s) + O_{2}(g)}$ $\Delta H_{2}'' = 2\times592 = +1184\ \text{kJ}$ (Wait, no, original Reaction 2 is $2SrO -> 2Sr + O2$, so maybe better to use Reaction 3 and the reversed Reaction 1 and Reaction 2.
Alternative approach: Let's express the target reaction in terms of the given reactions.
Target reaction: $\ce{C + O2 -> CO2}$
From Reaction 3: $\ce{SrCO3 -> Sr + C + 3/2 O2}$ $\Delta H_3' = +1220\ \text{kJ}$ (divided by 2)
From Reaction 1 reversed: $\ce{SrCO3 -> SrO + CO2}$ $\Delta H_1' = +234\ \text{kJ}$ (reversed, so $\Delta H = +234$; no multiplication yet)
From Reaction 2 divided: $\ce{SrO -> Sr + 1/2 O2}$ $\Delta H_2' = +592\ \text{kJ}$ (divided by 2)
Wait, maybe a better way: Let's use Hess's law. The target reaction is $\ce{C + O2 -> CO2}$. Let's find the sum of reactions to get this.
Let's take Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ $\Delta H3 = +2440$
Reverse Reaction 1 (times 2): $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H1' = +468$ (since original Reaction 1: $\ce{SrO + CO2 -> SrCO3}$ $\Delta H = -234$, so reversed is $\ce{SrCO3 -> SrO + CO2}$ $\Delta H = +234$, times 2: $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H = +468$)
Reverse Reaction 2: $\ce{2Sr + O2 -> 2SrO}$ $\Delta H2' = -1184$ (since original Reaction 2: $\ce{2SrO -> 2Sr + O2}$ $\Delta H = +1184$, reversed is $\ce{2Sr + O2 -> 2SrO}$ $\Delta H = -1184$)
Now, add Reaction 3, reversed Reaction 1 (times 2), and reversed Reaction 2:
Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ $\Delta H3 = +2440$
Reversed Reaction 1 (times 2): $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H1' = +468$ (Wait, no, reversed Reaction 1 is $\ce{SrCO3 -> SrO + CO2}$ $\Delta H = +234$, so times 2 is $\ce{2SrCO3 -> 2SrO + 2CO2}$ $\Delta H = +468$. But if we add Reaction 3 and reversed Reaction 1 (times 2), we get:
$\ce{2SrCO3 + 2SrCO3 -> 2Sr + 2C + 3O2 + 2SrO + 2CO2}$ No, that's not right. Let's correct.
Wait, the target reaction is $\ce{C + O2 -> CO2}$. Let's express each reaction in terms of C, O2, CO2.
From Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ $\Delta H3 = +2440$ → divide by 2: $\ce{SrCO3 -> Sr + C + 3/2 O2}$ $\Delta H3' = +1220$
From Reaction 1: $\ce{SrO + CO2 -> SrCO3}$ $\Delta H1 = -234$ → reversed: $\ce{SrCO3 -> SrO + CO2}$ $\Delta H1' = +234$
From Reaction 2: $\ce{2SrO -> 2Sr + O2}$ $\Delta H2 = +1184$ → divide by 2: $\ce{SrO -> Sr + 1/2 O2}$ $\Delta H2' = +592$
Now, substitute $\ce{SrCO3}$ from Reaction 3' into reversed Reaction 1':
$\ce{Sr + C + 3/2 O2 -> SrO + CO2}$ $\Delta H = 1220 + 234 = 1454$? No, wait, Reaction 3' is $\ce{SrCO3 -> Sr + C + 3/2 O2}$ (ΔH=+1220), reversed Reaction 1' is $\ce{SrCO3 -> SrO + CO2}$ (ΔH=+234). So if we set them equal: $\ce{Sr + C + 3/2 O2 -> SrO + CO2}$ (ΔH=1220 + 234? No, that's not correct. Hess's law is about adding reactions. Let's add Reaction 3' (divided by 2), reversed Reaction 1 (divided by 1), and reversed Reaction 2 (divided by 2).
Wait, let's list the reactions:
- $\ce{SrCO3 -> Sr + C + 3/2 O2}$ $\Delta H = +1220$ (Reaction 3 divided by 2)
- $\ce{SrCO3 -> SrO + CO2}$ $\Delta H = +234$ (Reaction 1 reversed)
- $\ce{SrO -> Sr + 1/2 O2}$ $\Delta H = +592$ (Reaction 2 divided by 2)
Now, subtract reaction 2 from reaction 1: (Reaction 1 - Reaction 2):
$\ce{SrCO3 - SrCO3 -> (Sr + C + 3/2 O2) - (SrO + CO2)}$ → $\ce{0 -> Sr + C + 3/2 O2 - SrO - CO2}$ → $\ce{SrO -> Sr + C + 3/2 O2 - CO2}$
Then add Reaction 3: $\ce{SrO -> Sr + 1/2 O2}$ (ΔH=+592)
So:
$\ce{SrO + SrO -> (Sr + C + 3/2 O2 - CO2) + (Sr + 1/2 O2)}$ → No, this is getting confusing. Let's use the first method with multiplying and reversing.
Let's define the target reaction as R: $\ce{C + O2 -> CO2}$ ΔH = ?
We can write R as:
From Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ (ΔH3=+2440)
Reverse Reaction 1 (times 2): $\ce{2SrCO3 -> 2SrO + 2CO2}$ (ΔH1'=+468)
Reverse Reaction 2: $\ce{2Sr + O2 -> 2SrO}$ (ΔH2'=-1184)
Now, add these three reactions:
Reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ (ΔH=+2440)
Reverse Reaction 1 (times 2): $\ce{2SrCO3 -> 2SrO + 2CO2}$ (ΔH=+468) → Wait, no, we can't add two reactions with 2SrCO3 on the left. Instead, subtract Reverse Reaction 1 (times 2) from Reaction 3:
Reaction 3 - Reverse Reaction 1 (times 2):
$\ce{2SrCO3 - 2SrCO3 -> 2Sr + 2C + 3O2 - (2SrO + 2CO2)}$ → $\ce{0 -> 2Sr + 2C + 3O2 - 2SrO - 2CO2}$ → $\ce{2SrO -> 2Sr + 2C + 3O2 - 2CO2}$
Now add Reverse Reaction 2: $\ce{2Sr + O2 -> 2SrO}$ (ΔH=-1184)
So:
$\ce{2SrO + 2Sr + O2 -> 2Sr + 2C + 3O2 - 2CO2 + 2SrO}$
Simplify:
$\ce{O2 -> 2C + 2O2 - 2CO2}$
$\ce{0 -> 2C + O2 - 2CO2}$
$\ce{2CO2 -> 2C + 2O2}$
Divide by 2:
$\ce{CO2 -> C + O2}$ ΔH = (2440 + 468 - 1184)/2? Wait, no, let's calculate the ΔH for the sum:
Reaction 3: +2440
Reverse Reaction 1 (times 2): +468
Reverse Reaction 2: -1184
Total ΔH for the sum: 2440 + 468 - 1184 = 1724
But the sum of the reactions is $\ce{2CO2 -> 2C + 2O2}$ (since we had 2SrCO3 - 2SrCO3 = 0, 2Sr - 2Sr = 0, 3O2 + O2 = 4O2, 2C remains, and -2CO2, so 2CO2 -> 2C + 2O2)
So ΔH for $\ce{2CO2 -> 2C + 2O2}$ is 1724 kJ
Therefore, ΔH for $\ce{CO2 -> C + O2}$ is 1724/2 = 862 kJ
But the target reaction is $\ce{C + O2 -> CO2}$, which is the reverse, so ΔH = -862? No, that can't be right. Wait, I must have made a mistake.
Wait, let's start over.
Target reaction: $\ce{C(s, gr) + O_{2}(g) -> CO_{2}(g)}$ ΔH = ?
Let's use Hess's law. Let's express the target reaction as a combination of the given reactions.
Given reactions:
- $\ce{SrO + CO2 -> SrCO3}$ ΔH1 = -234 kJ
- $\ce{2SrO -> 2Sr + O2}$ ΔH2 = +1184 kJ
- $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ ΔH3 = +2440 kJ
We need to eliminate SrO, Sr, and SrCO3.
Let's solve for $\ce{CO2}$ from reaction 1: $\ce{CO2 -> SrCO3 - SrO}$
From reaction 2: $\ce{SrO -> Sr + 0.5 O2}$ ΔH2' = 1184/2 = 592 kJ
From reaction 3: $\ce{SrCO3 -> Sr + C + 1.5 O2}$ ΔH3' = 2440/2 = 1220 kJ
Now, substitute $\ce{SrCO3}$ from reaction 3' into $\ce{CO2}$:
$\ce{CO2 -> (Sr + C + 1.5 O2) - SrO}$
But from reaction 2', $\ce{SrO -> Sr + 0.5 O2}$, so $\ce{Sr -> SrO - 0.5 O2}$
Substitute $\ce{Sr}$ into $\ce{CO2}$:
$\ce{CO2 -> (SrO - 0.5 O2 + C + 1.5 O2) - SrO}$
Simplify:
$\ce{CO2 -> SrO - 0.5 O2 + C + 1.5 O2 - SrO}$
$\ce{CO2 -> C + O2}$
Wait, that's the reverse of the target reaction. So $\ce{CO2 -> C + O2}$ ΔH = (ΔH3' - ΔH1 - ΔH2')
ΔH3' = 1220, ΔH1 = -234 (since reaction 1 is $\ce{SrO + CO2 -> SrCO3}$ ΔH=-234, so $\ce{CO2 -> SrCO3 - SrO}$ ΔH=-234), ΔH2' = 592
So ΔH for $\ce{CO2 -> C + O2}$ is 1220 - (-234) - 592 = 1220 + 234 - 592 = 862 kJ
Therefore, ΔH for $\ce{C + O2 -> CO2}$ is -862 kJ? But that's not correct, because the standard enthalpy of formation of CO2 is -393.5 kJ/mol. Wait, maybe the given reactions have different values, so let's check the calculations again.
Wait, reaction 3: $\ce{2SrCO3 -> 2Sr + 2C + 3O2}$ ΔH=+2440 kJ. So per mole of SrCO3, ΔH=1220 kJ.
Reaction 1: $\ce{SrO + CO2 -> SrCO3}$ ΔH=-234 kJ. So ΔH for $\ce{CO2 -> SrCO3 - SrO}$ is -234 kJ.
Reaction 2: $\ce{2SrO -> 2Sr + O2}$ ΔH=+1184 kJ. So ΔH for $\ce{SrO -> Sr + 0.5 O2}$ is 592 kJ.
Now, substitute $\ce{SrCO3}$ from reaction 3' into reaction 1:
$\ce{CO2 -> (Sr + C + 1.5 O2) - SrO}$
Now, substitute $\ce{SrO}$ from reaction 2':
$\ce{CO2 -> (Sr + C + 1.5 O2) - (Sr + 0.5 O2)}$
Simplify:
$\ce{CO2 -> Sr + C + 1.5 O2 - Sr - 0.5 O2}$
$\ce{CO2 -> C + O2}$
So ΔH for $\ce{CO2 -> C + O2}$ is ΔH3' - ΔH1 - ΔH