QUESTION IMAGE
Question
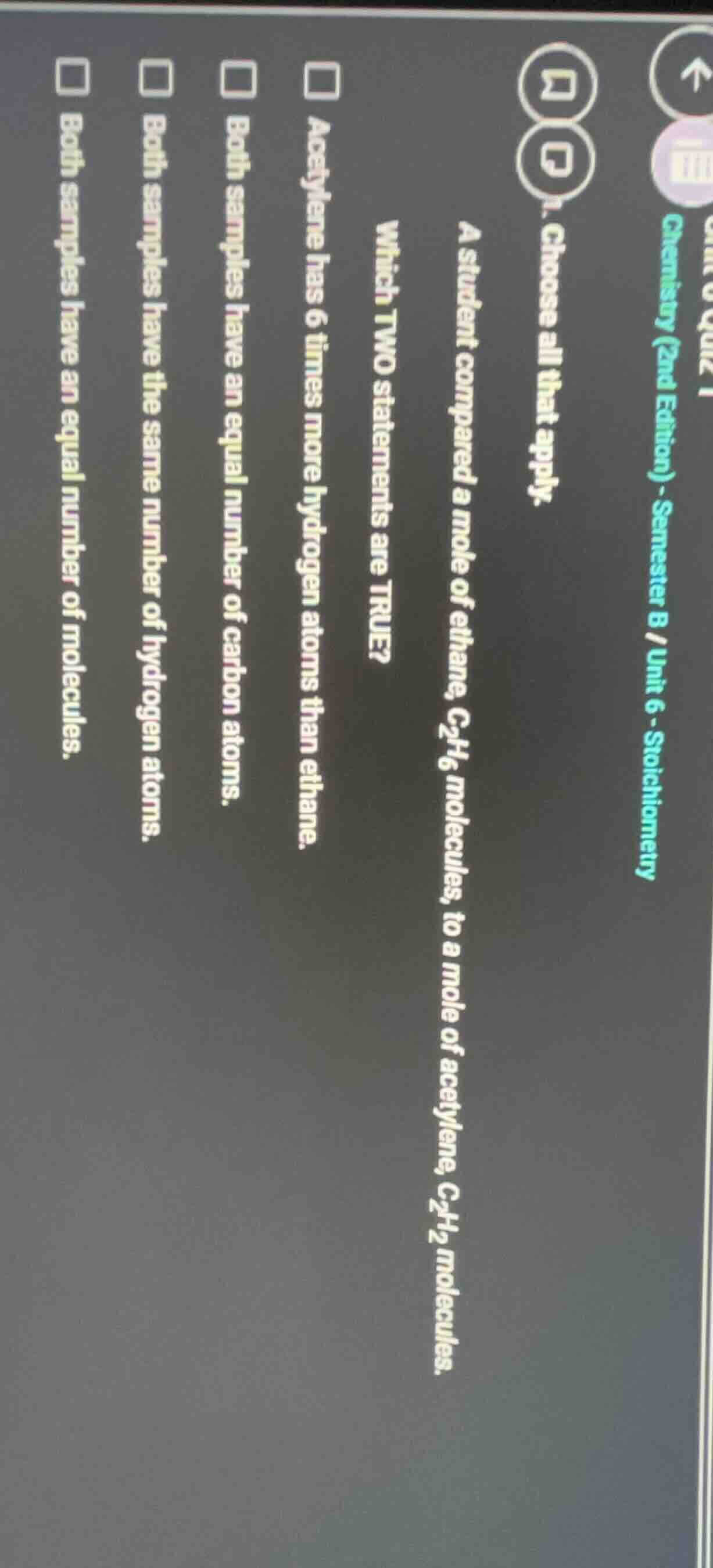
a student compared a mole of ethane, c₂h₆ molecules, to a mole of acetylene, c₂h₂ molecules. which two statements are true? acetylene has 6 times more hydrogen atoms than ethane. both samples have an equal number of carbon atoms. both samples have the same number of hydrogen atoms. both samples have an equal number of molecules. choose all that apply.
Step1: Molecule count comparison
1 mole of any substance contains $6.022 \times 10^{23}$ molecules, so 1 mole of $\text{C}_2\text{H}_6$ and 1 mole of $\text{C}_2\text{H}_2$ have equal molecule counts.
Step2: Carbon atom count comparison
1 molecule of $\text{C}_2\text{H}_6$ has 2 C atoms; 1 mole has $2 \times 6.022 \times 10^{23}$ C atoms. 1 molecule of $\text{C}_2\text{H}_2$ has 2 C atoms; 1 mole has $2 \times 6.022 \times 10^{23}$ C atoms. So equal C atoms.
Step3: Hydrogen atom count comparison
1 mole of $\text{C}_2\text{H}_6$ has $6 \times 6.022 \times 10^{23}$ H atoms; 1 mole of $\text{C}_2\text{H}_2$ has $2 \times 6.022 \times 10^{23}$ H atoms. Ethane has 3x more H atoms, not the reverse.
Step4: Verify correct statements
The true statements are equal molecules and equal carbon atoms.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Both samples have an equal number of carbon atoms.
- Both samples have an equal number of molecules.