QUESTION IMAGE
Question
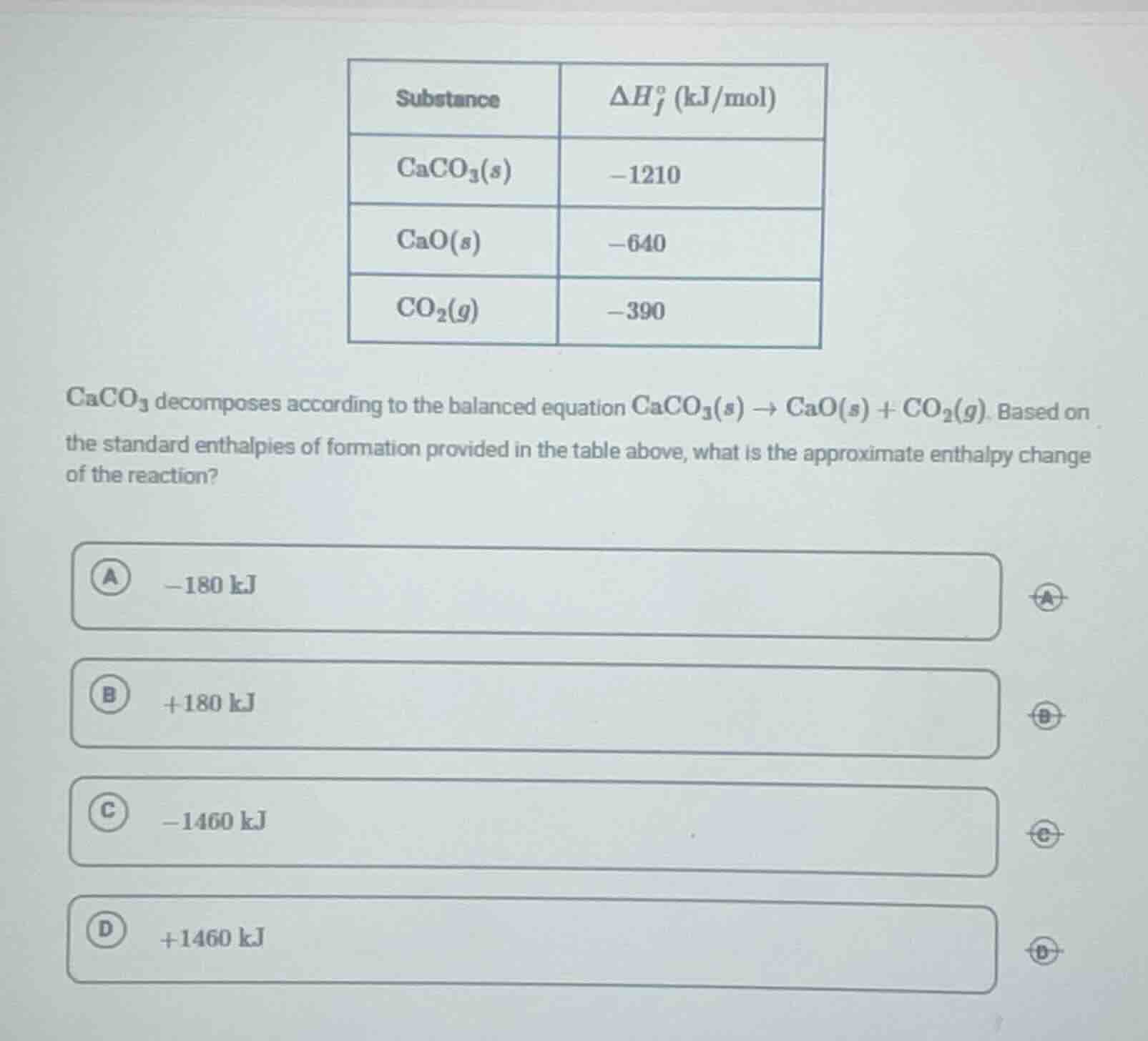
substance | $delta h_f^o$ (kj/mol)
caco₃(s) | -1210
cao(s) | -640
co₂(g) | -390
caco₃ decomposes according to the balanced equation caco₃(s) → cao(s) + co₂(g). based on the standard enthalpies of formation provided in the table above, what is the approximate enthalpy change of the reaction?
a -180 kj
b +180 kj
c -1460 kj
d +1460 kj
Step1: Recall the formula for enthalpy change of reaction
The enthalpy change of a reaction ($\Delta H_{rxn}$) is calculated by the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. The formula is:
$\Delta H_{rxn} = \sum \Delta H_f^{\circ}(\text{products}) - \sum \Delta H_f^{\circ}(\text{reactants})$
Step2: Identify products and reactants
For the reaction $\ce{CaCO_3(s) -> CaO(s) + CO_2(g)}$:
- Reactant: $\ce{CaCO_3(s)}$ with $\Delta H_f^{\circ} = -1210\ \text{kJ/mol}$
- Products: $\ce{CaO(s)}$ with $\Delta H_f^{\circ} = -640\ \text{kJ/mol}$ and $\ce{CO_2(g)}$ with $\Delta H_f^{\circ} = -390\ \text{kJ/mol}$
Step3: Calculate sum of products' $\Delta H_f^{\circ}$
Sum of products: $\Delta H_f^{\circ}(\ce{CaO}) + \Delta H_f^{\circ}(\ce{CO_2}) = (-640) + (-390) = -1030\ \text{kJ/mol}$
Step4: Calculate $\Delta H_{rxn}$
Using the formula:
$\Delta H_{rxn} = [\Delta H_f^{\circ}(\ce{CaO}) + \Delta H_f^{\circ}(\ce{CO_2})] - \Delta H_f^{\circ}(\ce{CaCO_3})$
Substitute the values:
$\Delta H_{rxn} = (-1030) - (-1210) = -1030 + 1210 = 180\ \text{kJ/mol}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. +180 kJ