QUESTION IMAGE
Question
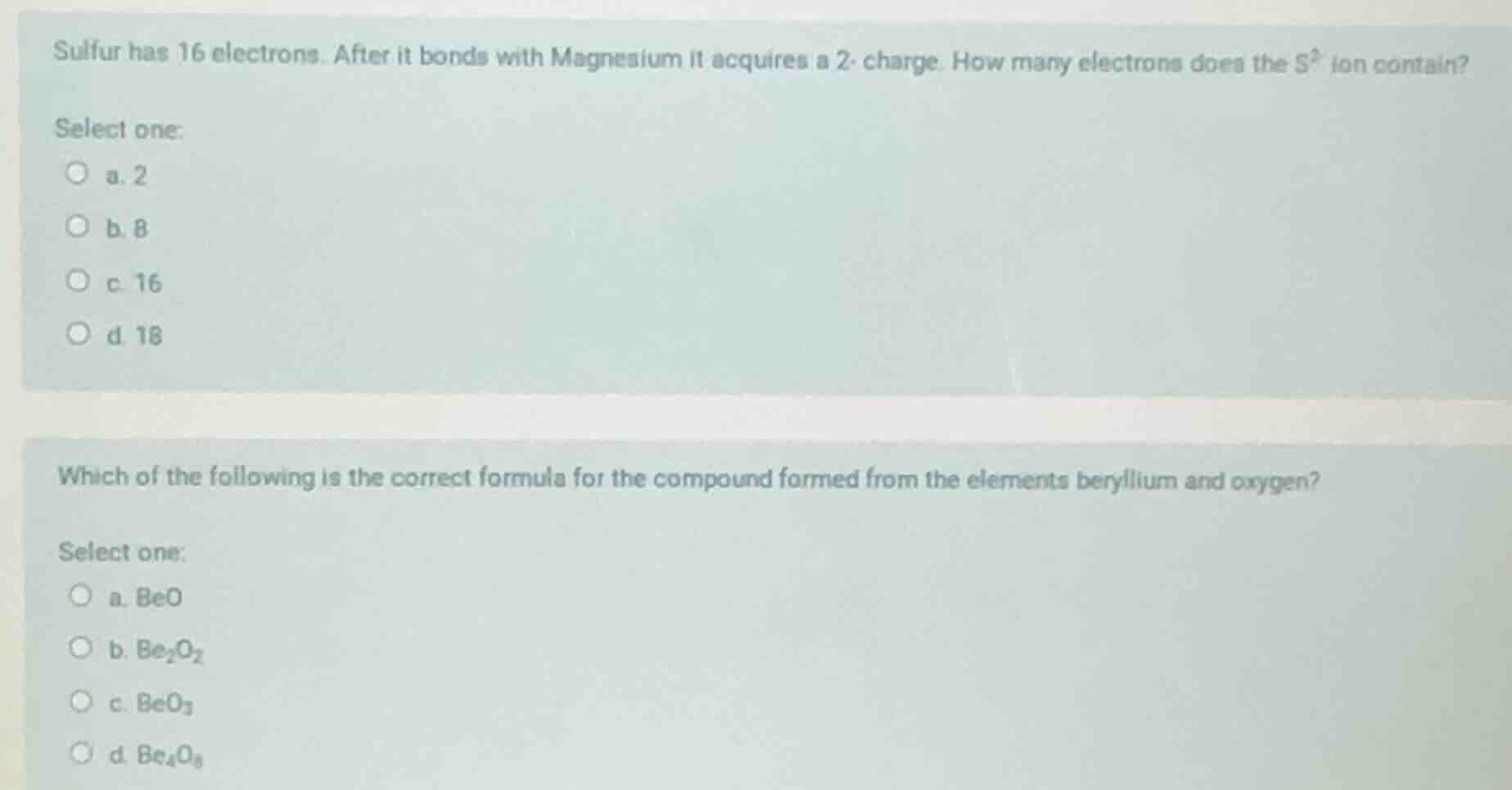
sulfur has 16 electrons. after it bonds with magnesium it acquires a 2- charge. how many electrons does the s²⁻ ion contain?
select one:
a. 2
b. 8
c. 16
d. 18
which of the following is the correct formula for the compound formed from the elements beryllium and oxygen?
select one:
a. beo
b. be₂o₂
c. beo₃
d. be₄o₈
Step1: Calculate S²⁻ electron count
A neutral S atom has 16 electrons. A 2⁻ charge means it gains 2 electrons:
$16 + 2 = 18$
Step2: Find Be-O compound formula
Beryllium forms $Be^{2+}$, oxygen forms $O^{2-}$. Charges balance in a 1:1 ratio, so the formula is $BeO$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- d. 18
- a. BeO