QUESTION IMAGE
Question
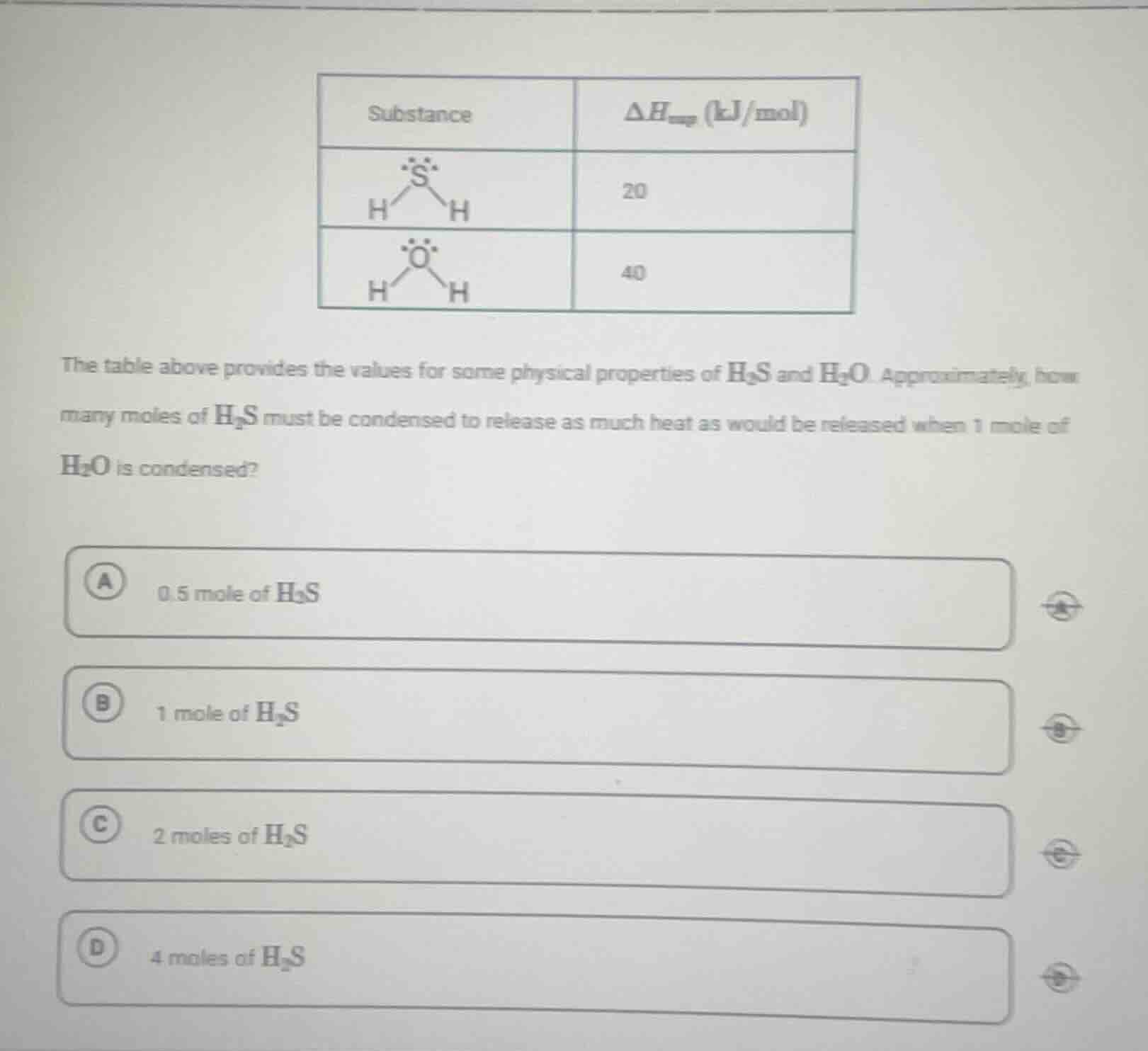
the table above provides the values for some physical properties of $\ce{h_{2}s}$ and $\ce{h_{2}o}$. approximately, how many moles of $\ce{h_{2}s}$ must be condensed to release as much heat as would be released when 1 mole of $\ce{h_{2}o}$ is condensed?
a 0.5 mole of $\ce{h_{2}s}$
b 1 mole of $\ce{h_{2}s}$
c 2 moles of $\ce{h_{2}s}$
d 4 moles of $\ce{h_{2}s}$
Step1: Understand heat release in condensation
Condensation releases heat equal to the enthalpy of vaporization ($\Delta H_{\text{vap}}$) but with opposite sign (since vaporization absorbs heat, condensation releases it). For 1 mole of $\ce{H2O}$, heat released $Q_{\ce{H2O}} = 1\ \text{mol} \times 40\ \text{kJ/mol} = 40\ \text{kJ}$.
Step2: Let moles of $\ce{H2S}$ be $n$
Heat released by $n$ moles of $\ce{H2S}$ is $Q_{\ce{H2S}} = n \times 20\ \text{kJ/mol}$. We need $Q_{\ce{H2S}} = Q_{\ce{H2O}}$, so:
$$n \times 20 = 40$$
Step3: Solve for $n$
Divide both sides by 20: $n = \frac{40}{20} = 2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. 2 moles of $\ce{H2S}$