QUESTION IMAGE
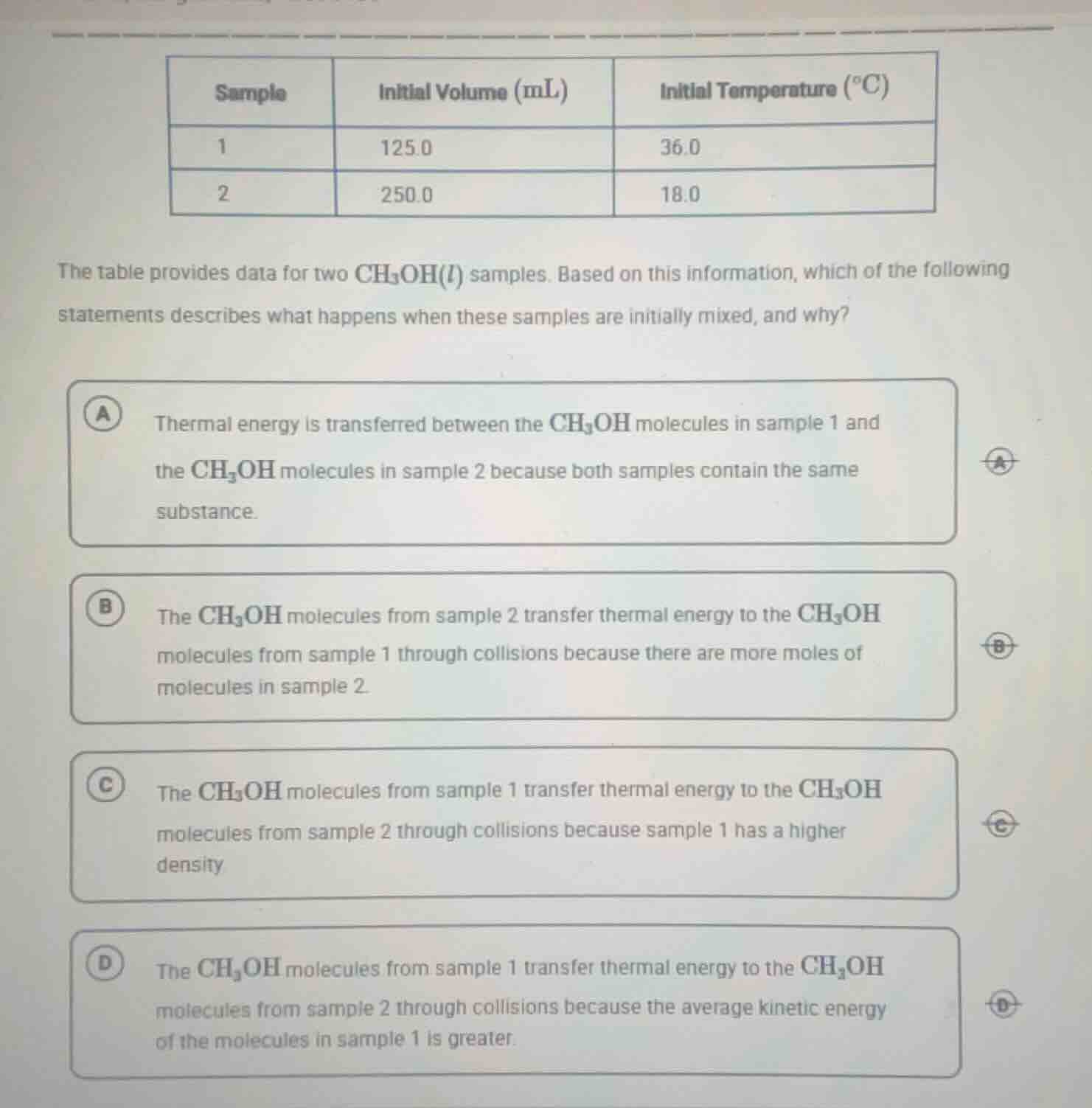
Question
the table provides data for two ch₃oh(l) samples. based on this information, which of the following statements describes what happens when these samples are initially mixed, and why?
a thermal energy is transferred between the ch₃oh molecules in sample 1 and the ch₃oh molecules in sample 2 because both samples contain the same substance.
b the ch₃oh molecules from sample 2 transfer thermal energy to the ch₃oh molecules from sample 1 through collisions because there are more moles of molecules in sample 2.
c the ch₃oh molecules from sample 1 transfer thermal energy to the ch₃oh molecules from sample 2 through collisions because sample 1 has a higher density
d the ch₃oh molecules from sample 1 transfer thermal energy to the ch₃oh molecules from sample 2 through collisions because the average kinetic energy of the molecules in sample 1 is greater.
- Option A: Thermal energy transfer between same - substance samples is not solely because they are the same substance. The key factor is temperature difference, so A is incorrect.
- Option B: The number of moles (related to volume here, but since it's the same substance, density is constant) is not the reason for thermal energy transfer. Thermal energy transfer is due to temperature difference (average kinetic energy), so B is incorrect.
- Option C: Density of the same substance (liquid methanol) is constant. Thermal energy transfer is not related to density but to temperature (average kinetic energy) difference, so C is incorrect.
- Option D: The temperature of sample 1 ($36.0^{\circ}C$) is higher than that of sample 2 ($18.0^{\circ}C$). The average kinetic energy of molecules is related to temperature (higher temperature means higher average kinetic energy). When two samples are mixed, molecules with higher average kinetic energy (from sample 1) will transfer thermal energy to molecules with lower average kinetic energy (from sample 2) through collisions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. The $\ce{CH_{3}OH}$ molecules from sample 1 transfer thermal energy to the $\ce{CH_{3}OH}$ molecules from sample 2 through collisions because the average kinetic energy of the molecules in sample 1 is greater.