QUESTION IMAGE
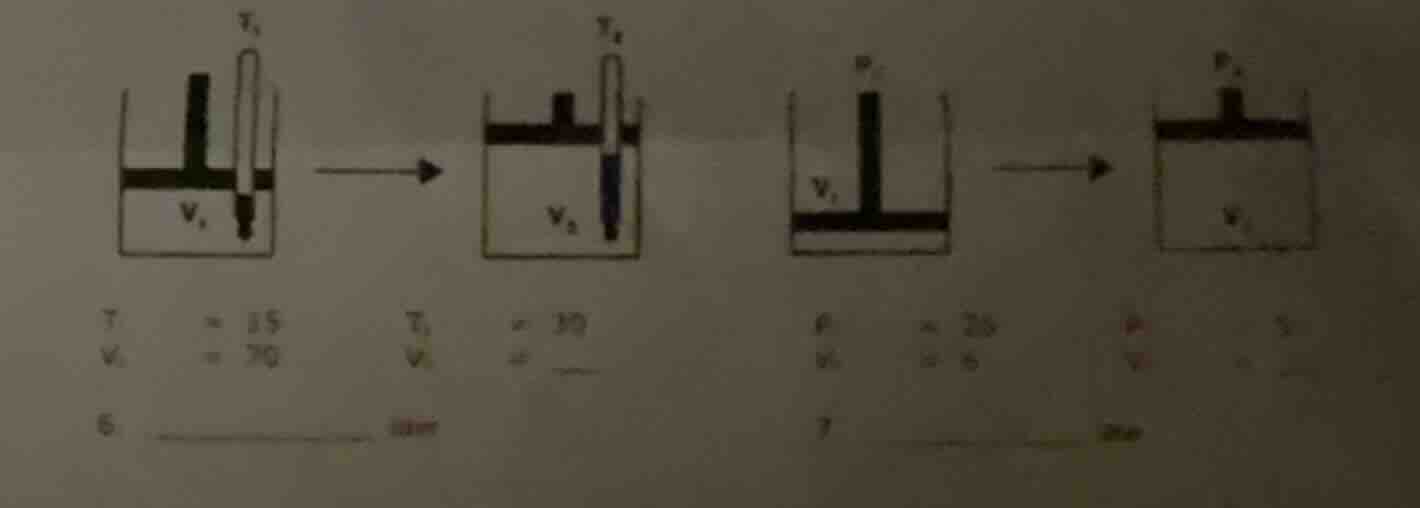
Question
there are four figures showing different states of gas containers with thermometers and pressure - related setups. below the figures, there are some values: for the first part, t₁ = 15, v₁ = 70; t₂ = 30, v₂ = __. for the second part, p₁ = 20, v₁ = 5; p₂ = 5, v₂ = . and there are labels 6. law and 7. __ law.
Problem 6 (Relating to Charles's Law, part of Physics in Natural Science)
Step 1: Recall Charles's Law
Charles's Law states that for a fixed amount of gas at constant pressure, $\frac{V_1}{T_1}=\frac{V_2}{T_2}$, where $V$ is volume and $T$ is absolute temperature (in Kelvin, but if we assume the temperatures here are in a consistent scale like Celsius for proportionality, we can use the formula directly). Given $T_1 = 15$, $V_1=70$, $T_2 = 30$. We need to find $V_2$.
Step 2: Apply Charles's Law formula
From $\frac{V_1}{T_1}=\frac{V_2}{T_2}$, we can re - arrange for $V_2$: $V_2=\frac{V_1\times T_2}{T_1}$
Substitute the values: $V_1 = 70$, $T_1=15$, $T_2 = 30$
$V_2=\frac{70\times30}{15}$
Step 3: Calculate the value
First, calculate $70\times30 = 2100$, then divide by 15: $\frac{2100}{15}=140$
Step 1: Recall Boyle's Law
Boyle's Law states that for a fixed amount of gas at constant temperature, $P_1V_1=P_2V_2$, where $P$ is pressure and $V$ is volume. Given $P_1 = 20$, $V_1 = 5$, $P_2=5$. We need to find $V_2$.
Step 2: Apply Boyle's Law formula
From $P_1V_1 = P_2V_2$, we can re - arrange for $V_2$: $V_2=\frac{P_1V_1}{P_2}$
Substitute the values: $P_1 = 20$, $V_1=5$, $P_2 = 5$
$V_2=\frac{20\times5}{5}$
Step 3: Calculate the value
First, calculate $20\times5=100$, then divide by 5: $\frac{100}{5} = 20$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$V_2 = 140$