QUESTION IMAGE
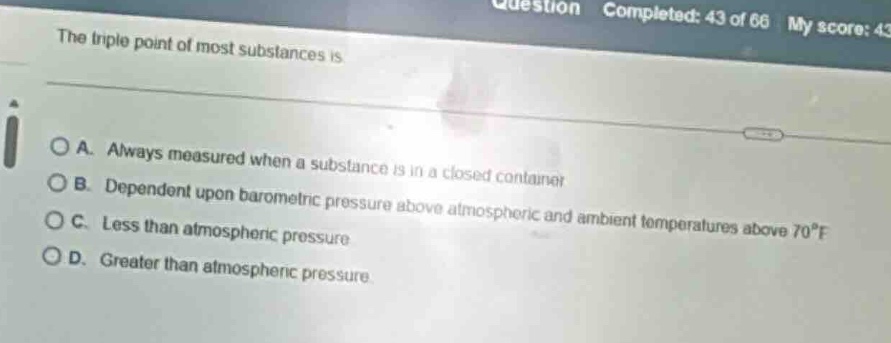
Question
the triple point of most substances is
a. always measured when a substance is in a closed container
b. dependent upon barometric pressure above atmospheric and ambient temperatures above 70°f
c. less than atmospheric pressure
d. greater than atmospheric pressure.
The triple point is a fixed temperature-pressure pair where a substance's solid, liquid, and gas phases coexist in equilibrium. For most substances, this pressure is lower than standard atmospheric pressure (1 atm = 101.325 kPa). Option A is incorrect because the triple point is an intrinsic property, not dependent on measurement container type. Option B is wrong as the triple point is a fixed value, not dependent on external barometric pressure or ambient temperatures. Option D is incorrect, as only a few substances (like water) have a triple point pressure near atmospheric, and most are lower.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Less than atmospheric pressure