QUESTION IMAGE
Question
try it
reactions
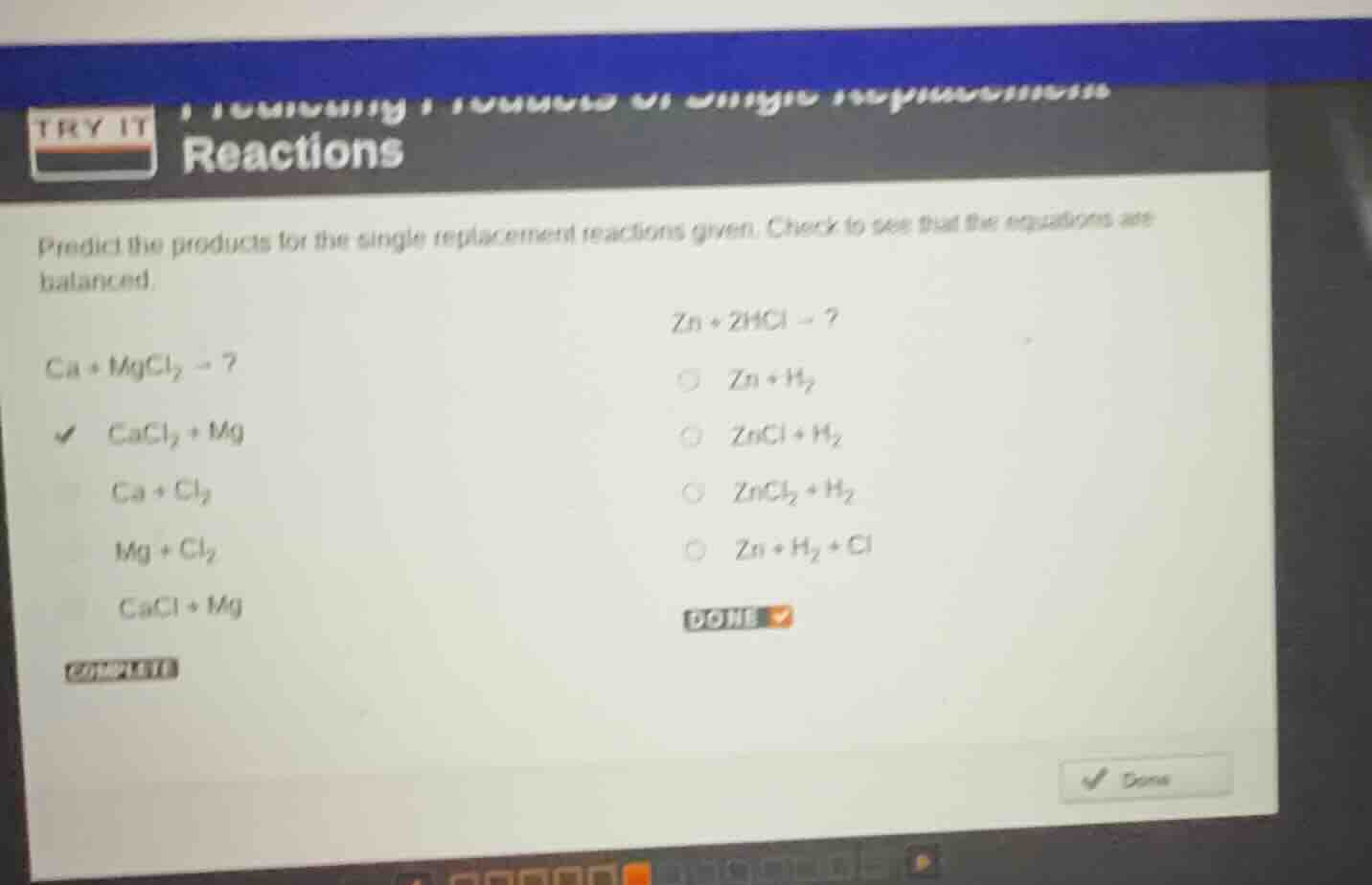
predict the products for the single replacement reactions given. check to see that the equations are balanced.
$ca + mgcl_2 \
ightarrow ?$
$cacl_2 + mg$
$ca + cl_2$
$mg + cl_2$
$cacl + mg$
$zn + 2hcl \
ightarrow ?$
$zn + h_2$
$zncl + h_2$
$zncl_2 + h_2$
$zn + h_2 + cl$
complete
done
Step1: Identify single replacement pattern
In single replacement, a metal replaces the cation in an ionic acid/salt. For $\text{Zn} + 2\text{HCl}$, Zn replaces H in HCl.
Step2: Write unbalanced product equation
$\text{Zn} + 2\text{HCl}
ightarrow \text{ZnCl}_2 + \text{H}_2$
Step3: Verify balanced equation
Count atoms: Zn: 1 on both sides; H: 2 on both sides; Cl: 2 on both sides. The equation is balanced.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\text{ZnCl}_2 + \text{H}_2$