QUESTION IMAGE
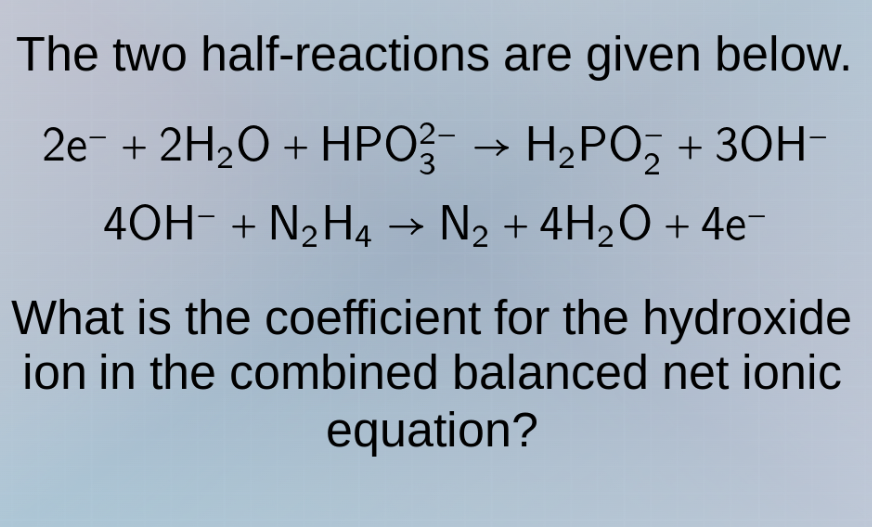
Question
the two half - reactions are given below.
$2e^- + 2h_2o + hpo_3^{2-} \
ightarrow h_2po_2^- + 3oh^-$
$4oh^- + n_2h_4 \
ightarrow n_2 + 4h_2o + 4e^-$
what is the coefficient for the hydroxide ion in the combined balanced net ionic equation?
Response
again.
For the first half - reaction (multiplied by 2):
- P: 2 on left, 2 on right (good)
- O: \(2\times3 + 4\times1=6 + 4 = 10\) on left (2 \(HPO_3^{2 - }\) has 6 O, 4 \(H_2O\) has 4 O); on right, 2\times2+6\times1 = 4 + 6=10 O (2 \(H_2PO_2^-\) has 4 O, 6 \(OH^-\) has 6 O) (good)
- H: 4\times2 + 2\times2=8 + 4 = 12? Wait, no, \(2HPO_3^{2 - }\) has 2 H, \(4H_2O\) has 8 H, total 10 H. On right, 2\times2+6\times1=4 + 6 = 10 H (2 \(H_2PO_2^-\) has 4 H, 6 \(OH^-\) has 6 H) (good)
Second half - reaction:
- N: 2 on left, 2 on right (good)
- H: 4 on left, 4\times2 = 8 on right? Wait, no, \(N_2H_4\) has 4 H, \(4H_2O\) has 8 H? No, \(4H_2O\) has 8 H? Wait, \(N_2H_4
ightarrow4H_2O\): \(N_2H_4\) has 4 H, \(4H_2O\) has 8 H.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
again.
For the first half - reaction (multiplied by 2):
- P: 2 on left, 2 on right (good)
- O: \(2\times3 + 4\times1=6 + 4 = 10\) on left (2 \(HPO_3^{2 - }\) has 6 O, 4 \(H_2O\) has 4 O); on right, 2\times2+6\times1 = 4 + 6=10 O (2 \(H_2PO_2^-\) has 4 O, 6 \(OH^-\) has 6 O) (good)
- H: 4\times2 + 2\times2=8 + 4 = 12? Wait, no, \(2HPO_3^{2 - }\) has 2 H, \(4H_2O\) has 8 H, total 10 H. On right, 2\times2+6\times1=4 + 6 = 10 H (2 \(H_2PO_2^-\) has 4 H, 6 \(OH^-\) has 6 H) (good)
Second half - reaction:
- N: 2 on left, 2 on right (good)
- H: 4 on left, 4\times2 = 8 on right? Wait, no, \(N_2H_4\) has 4 H, \(4H_2O\) has 8 H? No, \(4H_2O\) has 8 H? Wait, \(N_2H_4
ightarrow4H_2O\): \(N_2H_4\) has 4 H, \(4H_2O\) has 8 H.