QUESTION IMAGE
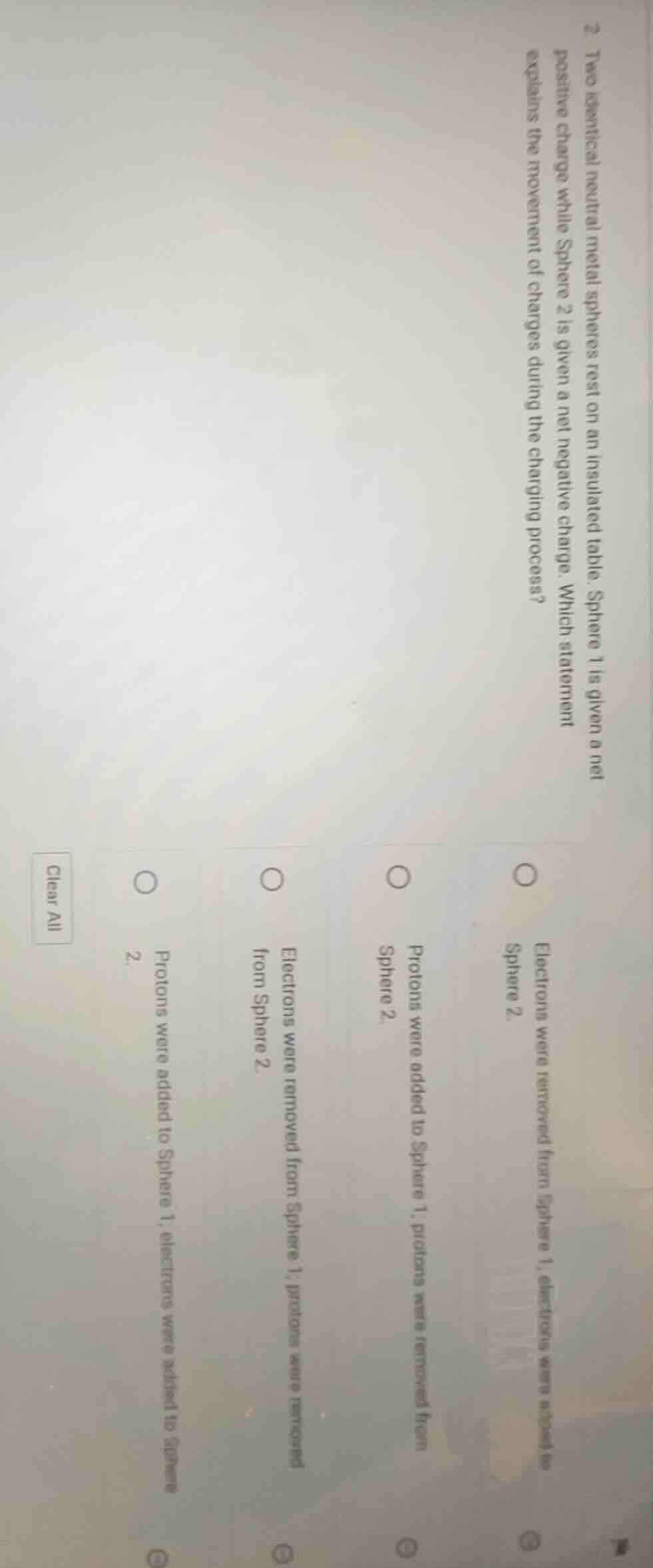
Question
- two identical neutral metal spheres rest on an insulated table. sphere 1 is given a net positive charge while sphere 2 is given a net negative charge. which statement explains the movement of charges during the charging process?○ electrons were removed from sphere 1, electrons were added to sphere 2.○ protons were added to sphere 1, protons were removed from sphere 2.○ electrons were removed from sphere 1, protons were removed from sphere 2.○ protons were added to sphere 1, electrons were added to sphere 2.clear all
Brief Explanations
In metallic spheres, protons are fixed in the nucleus and cannot move. A net positive charge forms when electrons are removed from the sphere, and a net negative charge forms when electrons are added to the sphere.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Electrons were removed from Sphere 1; electrons were added to Sphere 2.