QUESTION IMAGE
Question
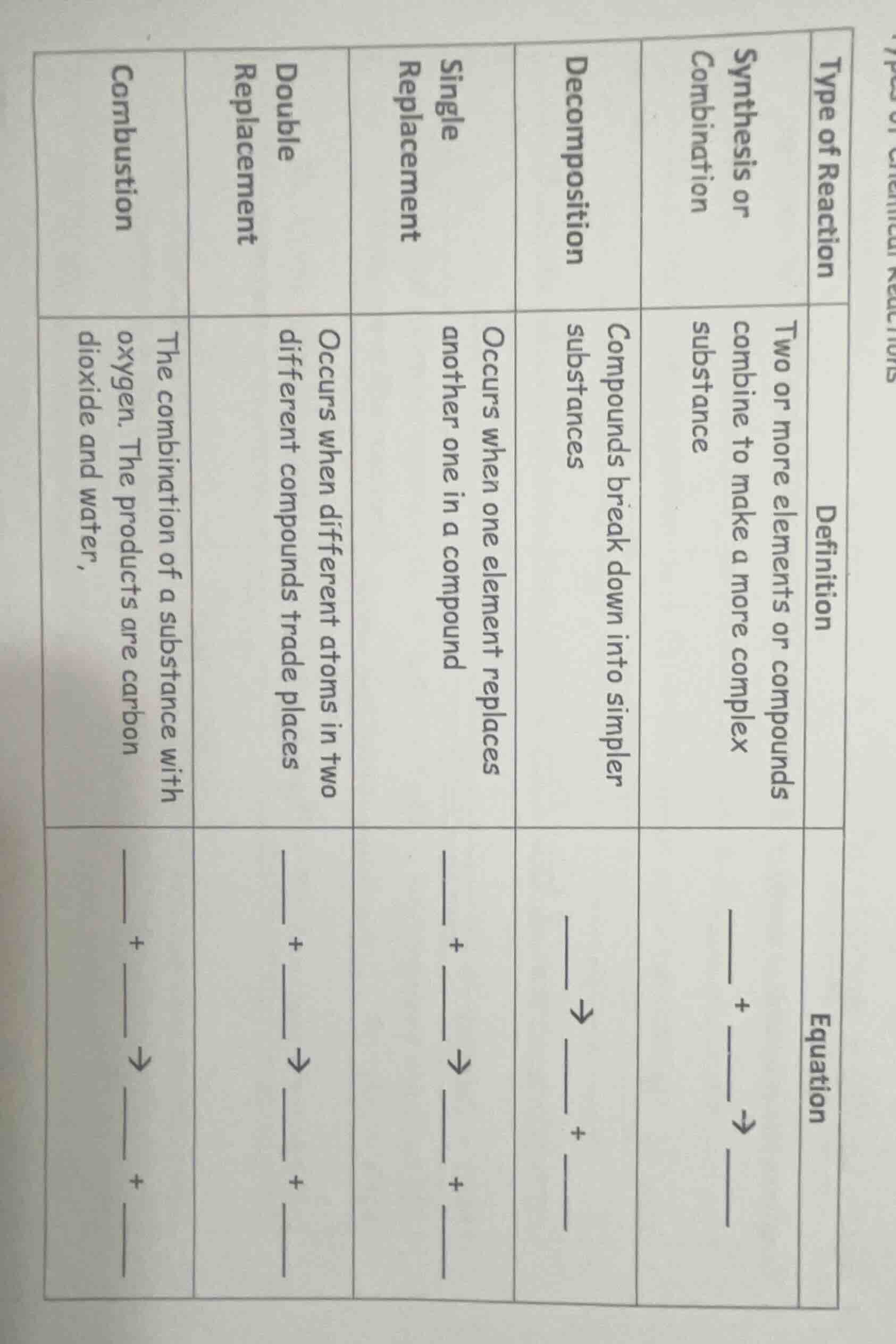
type of reaction
synthesis or combination
decomposition
single replacement
double replacement
combustion
definition
two or more elements or compounds combine to make a more complex substance
compounds break down into simpler substances
occurs when one element replaces another one in a compound
occurs when different atoms in two different compounds trade places
the combination of a substance with oxygen. the products are carbon dioxide and water,
equation
__ + → __
__ → + __
__ + → + __
__ + → + __
__ + → + __
Step1: Synthesis or Combination Reaction
A synthesis reaction is when two or more substances combine to form a single product. The general form is \( A + B
ightarrow AB \). So the equation should be filled as \( \text{Element/Compound 1} + \text{Element/Compound 2}
ightarrow \text{Compound} \). For example, \( \ce{H_2 + O_2
ightarrow H_2O} \) (after balancing), but in general form: \( A + B
ightarrow AB \).
Step2: Decomposition Reaction
A decomposition reaction is when a compound breaks down into simpler substances. The general form is \( AB
ightarrow A + B \). So the equation is \( \text{Compound}
ightarrow \text{Substance 1} + \text{Substance 2} \), e.g., \( \ce{H_2O_2
ightarrow H_2O + O_2} \) (after balancing), general form: \( AB
ightarrow A + B \).
Step3: Single Replacement Reaction
A single replacement reaction is when an element replaces another in a compound. The general form is \( A + BC
ightarrow AC + B \). So the equation is \( \text{Element} + \text{Compound}
ightarrow \text{New Compound} + \text{New Element} \), e.g., \( \ce{Zn + HCl
ightarrow ZnCl_2 + H_2} \) (after balancing), general form: \( A + BC
ightarrow AC + B \).
Step4: Double Replacement Reaction
A double replacement reaction is when two compounds exchange ions. The general form is \( AB + CD
ightarrow AD + CB \). So the equation is \( \text{Compound 1} + \text{Compound 2}
ightarrow \text{Compound 3} + \text{Compound 4} \), e.g., \( \ce{AgNO_3 + NaCl
ightarrow AgCl + NaNO_3} \), general form: \( AB + CD
ightarrow AD + CB \).
Step5: Combustion Reaction
A combustion reaction involves a substance reacting with oxygen, producing carbon dioxide and water (for hydrocarbon combustion). The general form for a hydrocarbon ( \( C_xH_y \)) is \( \ce{C_xH_y + O_2
ightarrow CO_2 + H_2O} \) (after balancing). So the equation is \( \text{Substance (e.g., Hydrocarbon)} + \ce{O_2}
ightarrow \ce{CO_2} + \ce{H_2O} \), e.g., \( \ce{CH_4 + O_2
ightarrow CO_2 + H_2O} \) (after balancing).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Synthesis or Combination: \( A + B
ightarrow AB \) (where \( A, B \) are elements/compounds, \( AB \) is a compound)
- Decomposition: \( AB
ightarrow A + B \) (where \( AB \) is a compound, \( A, B \) are simpler substances)
- Single Replacement: \( A + BC
ightarrow AC + B \) (where \( A \) is an element, \( BC \) is a compound, \( AC \) is a new compound, \( B \) is a new element)
- Double Replacement: \( AB + CD
ightarrow AD + CB \) (where \( AB, CD \) are compounds, \( AD, CB \) are new compounds)
- Combustion: \( \ce{C_xH_y + O_2
ightarrow CO_2 + H_2O} \) (for hydrocarbon combustion, general form; for other substances, \( \text{Substance} + \ce{O_2}
ightarrow \text{Oxide(s)} + \text{Other Products} \))
(Note: These are general chemical equation formats for each reaction type. Specific examples would require balancing and specific reactants/products, but the general forms are as above.)