QUESTION IMAGE
Question
types of reactions
quiz complete
1 2 3
time remaining
49:03
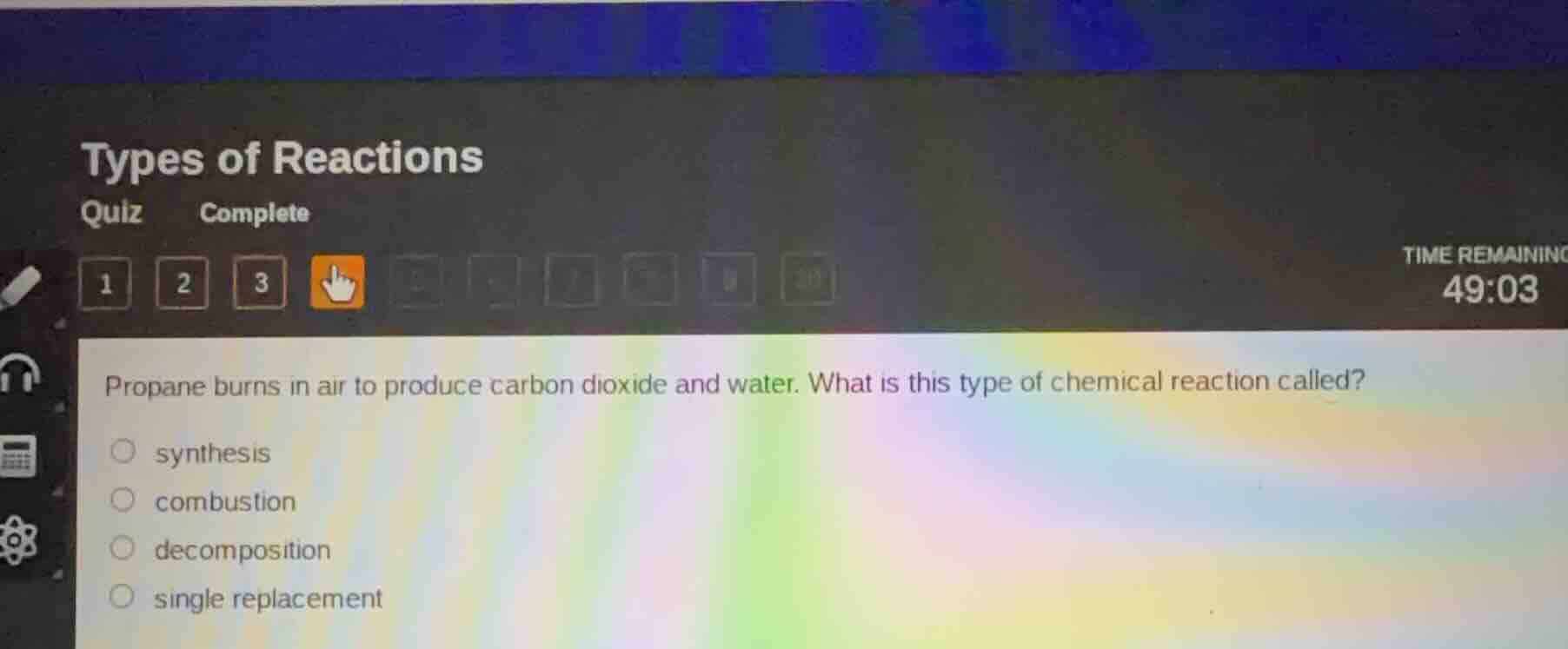
propane burns in air to produce carbon dioxide and water. what is this type of chemical reaction called?
○ synthesis
○ combustion
○ decomposition
○ single replacement
A combustion reaction involves a fuel reacting with oxygen (from air) to produce oxides (like carbon dioxide) and water, often with heat release. Propane (a hydrocarbon fuel) burning in air fits this definition. Synthesis combines simple substances into a complex one, decomposition breaks a compound into simpler ones, and single replacement involves one element replacing another in a compound, none of which match the described reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. combustion