QUESTION IMAGE
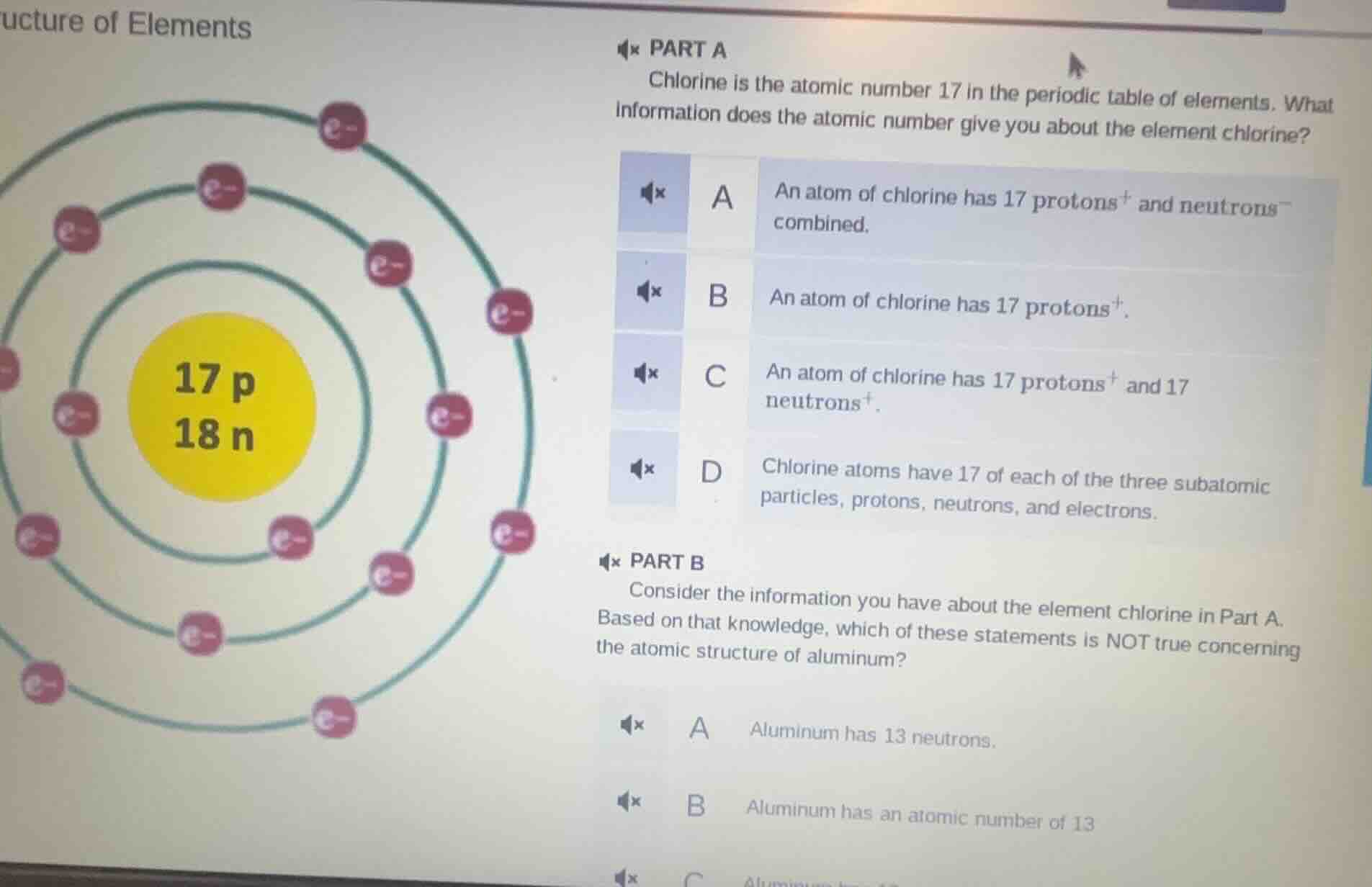
Question
ucture of elements
part a
chlorine is the atomic number 17 in the periodic table of elements. what
information does the atomic number give you about the element chlorine?
a an atom of chlorine has 17 protons⁺ and neutrons⁻ combined.
b an atom of chlorine has 17 protons⁺.
c an atom of chlorine has 17 protons⁺ and 17 neutrons⁺.
d chlorine atoms have 17 of each of the three subatomic particles, protons, neutrons, and electrons.
part b
consider the information you have about the element chlorine in part a.
based on that knowledge, which of these statements is not true concerning
the atomic structure of aluminum?
a aluminum has 13 neutrons.
b aluminum has an atomic number of 13
Step1: Analyze atomic number definition
The atomic number of an element is defined as the number of protons in the nucleus of its neutral atom.
Step2: Evaluate Part A options
- Option A: Incorrect. Protons + neutrons = mass number, not atomic number.
- Option B: Correct. Atomic number = number of protons.
- Option C: Incorrect. Neutrons are neutral, not positive; and atomic number does not equal neutron count.
- Option D: Incorrect. Atomic number only defines proton (and electron, in neutral atom) count, not neutron count.
Step3: Analyze Part B (incomplete, but use known data)
Aluminum has atomic number 13, so it has 13 protons. Its most common isotope has a mass number of 27, so neutrons = $27-13=14$.
- Option A: Incorrect (not true, as neutrons =14).
- Option B: Correct (aluminum's atomic number is 13).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Part A: B. An atom of chlorine has 17 protons⁺.
Part B: A. Aluminum has 13 neutrons.