QUESTION IMAGE
Question
unit 4 - reactions and enzymes
- define the following terms:
a. enzyme
b. catalyst
c. activation energy
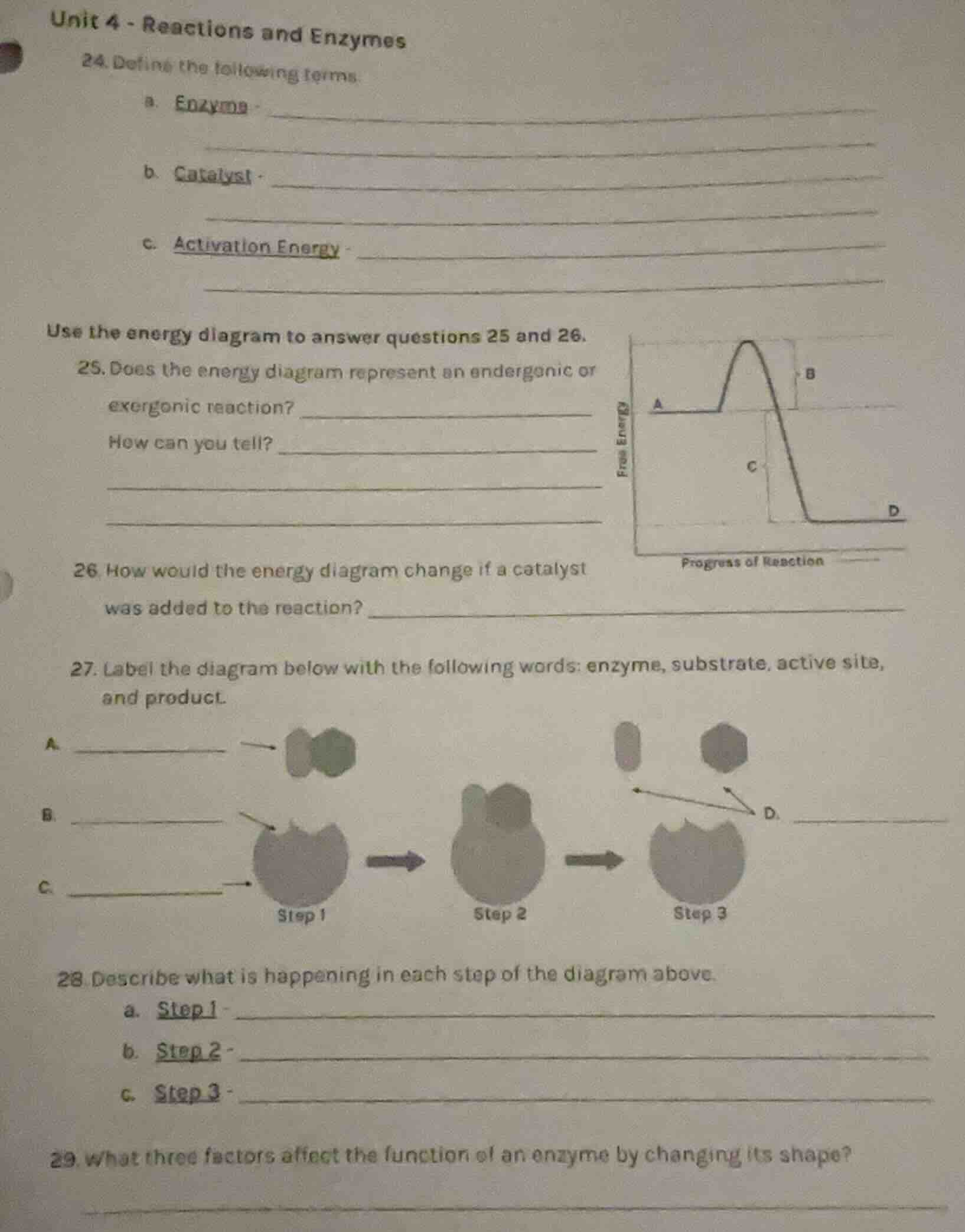
use the energy diagram to answer questions 25 and 26.
- does the energy diagram represent an endergonic or
exergonic reaction?
how can you tell?
- how would the energy diagram change if a catalyst
was added to the reaction?
- label the diagram below with the following words: enzyme, substrate, active site,
and product.
a.
b.
c.
d.
- describe what is happening in each step of the diagram above.
a. step 1
b. step 2
c. step 3
- what three factors affect the function of an enzyme by changing its shape?
Question 24
a. Enzyme
An enzyme is a biological catalyst, typically a protein (or sometimes RNA), that speeds up chemical reactions in living organisms by lowering the activation energy required for the reaction to occur. It is highly specific to its substrate (the molecule it acts on) and functions within a particular range of temperature and pH.
A catalyst is a substance that increases the rate of a chemical reaction without being consumed in the process. It works by lowering the activation energy of the reaction, allowing the reaction to proceed more quickly. Catalysts can be inorganic (like metals) or organic (like enzymes in biological systems).
Activation energy is the minimum amount of energy that reactant molecules must possess in order to undergo a chemical reaction. It is the energy barrier that must be overcome for the reaction to start. Catalysts (including enzymes) function by reducing this energy barrier.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A biological catalyst (usually a protein or RNA) that speeds up chemical reactions in cells by lowering activation energy, specific to its substrate.