QUESTION IMAGE
Question
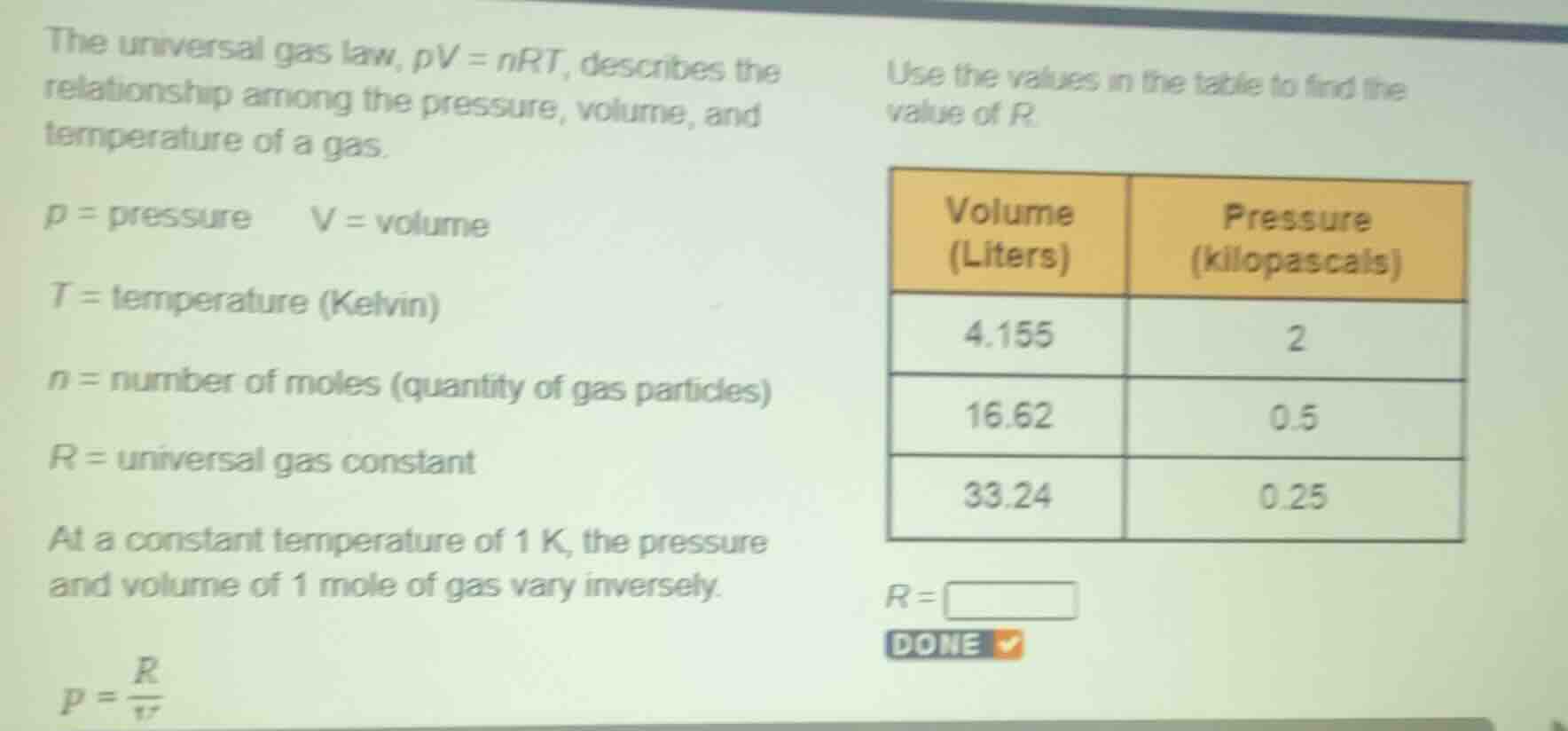
the universal gas law, $pv = nrt$, describes the relationship among the pressure, volume, and temperature of a gas.
$p = $ pressure $\quad v = $ volume
$t = $ temperature (kelvin)
$n = $ number of moles (quantity of gas particles)
$r = $ universal gas constant
at a constant temperature of 1 k, the pressure and volume of 1 mole of gas vary inversely.
$p = \frac{r}{v}$
use the values in the table to find the value of $r$.
| volume (liters) | pressure (kilopascals) |
|---|---|
| 16.62 | 0.5 |
| 33.24 | 0.25 |
$r = \square$
done
Step1: Rearrange gas law for $R$
From $pV = nRT$, solve for $R$:
$$R = \frac{pV}{nT}$$
Step2: Substitute given values
We know $n=1$ mole, $T=1$ K. Use first table row ($p=2$ kPa, $V=4.155$ L):
$$R = \frac{2 \times 4.155}{1 \times 1}$$
Step3: Calculate the value
$$R = 8.31$$
(Verify with other rows: $\frac{0.5 \times 16.62}{1 \times 1}=8.31$, $\frac{0.25 \times 33.24}{1 \times 1}=8.31$)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$8.31$ kilopascal-liters per mole-Kelvin ($\text{kPa·L/(mol·K)}$)