QUESTION IMAGE
Question
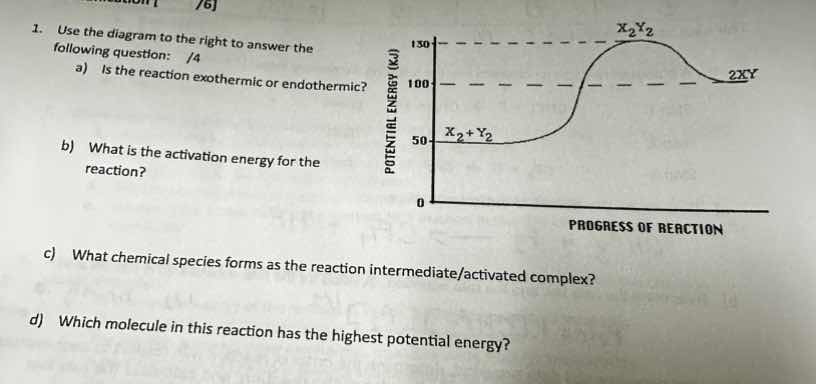
- use the diagram to the right to answer the following question: /4
a) is the reaction exothermic or endothermic?
b) what is the activation energy for the reaction?
c) what chemical species forms as the reaction intermediate/activated complex?
d) which molecule in this reaction has the highest potential energy?
Part (a)
Step1: Recall exothermic/endothermic definitions
Exothermic: products have less energy than reactants. Endothermic: products have more energy than reactants.
Step2: Compare reactant and product energy
Reactant (\(X_2 + Y_2\)) energy: 50 kJ. Product (\(2XY\)) energy: 100 kJ. Since product energy > reactant energy, the reaction is endothermic.
Step1: Recall activation energy definition
Activation energy (\(E_a\)) is the energy difference between reactants and the activated complex.
Step2: Calculate \(E_a\)
Reactant energy: 50 kJ. Activated complex (\(X_2Y_2\)) energy: 130 kJ. \(E_a = 130 - 50 = 80\) kJ.
Step1: Recall activated complex definition
The activated complex (reaction intermediate) is the high - energy species at the peak of the potential energy diagram.
Step2: Identify the species
From the diagram, the species at the peak is \(X_2Y_2\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Endothermic