QUESTION IMAGE
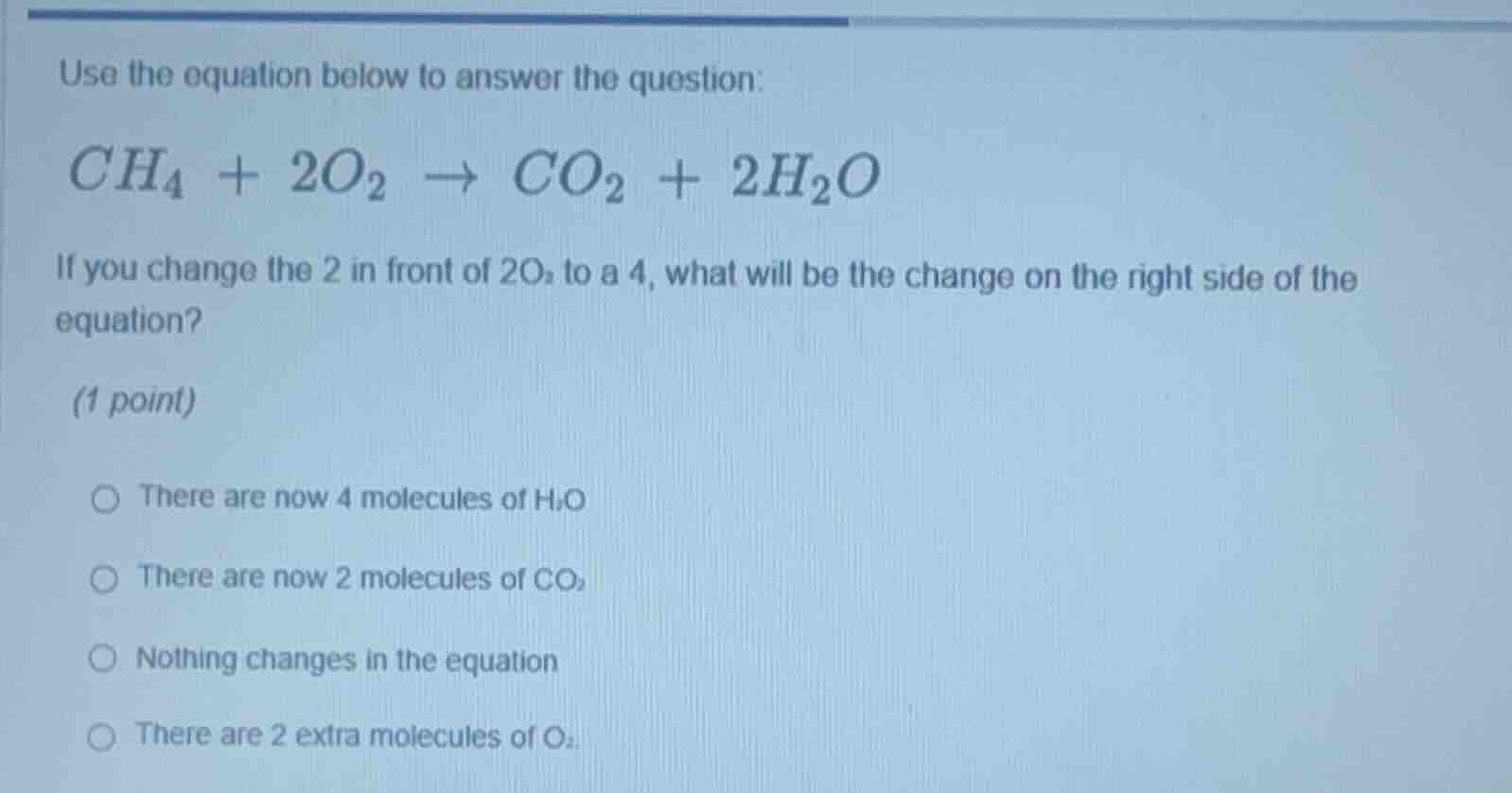
Question
use the equation below to answer the question: ch₄ + 2o₂ → co₂ + 2h₂o if you change the 2 in front of 2o₂ to a 4, what will be the change on the right side of the equation? (1 point) there are now 4 molecules of h₂o there are now 2 molecules of co₂ nothing changes in the equation there are 2 extra molecules of o₂
Step1: Analyze the original equation
The original equation is \( CH_4 + 2O_2
ightarrow CO_2 + 2H_2O \), which is balanced for atoms of C, H, and O.
Step2: Analyze the change in reactants
When we change the coefficient of \( O_2 \) from 2 to 4, the number of O atoms on the left side doubles (from \( 2\times2 = 4 \) to \( 4\times2 = 8 \) O atoms).
Step3: Determine the change in products to balance O
To balance the O atoms on the right side, we need to adjust the coefficients of \( CO_2 \) and \( H_2O \). Let's first look at the H atoms. The number of H atoms from \( CH_4 \) is 4, so the coefficient of \( H_2O \) should be such that \( 2\times n(H_2O)=4 \), but if O is increased, we also need to balance O. Wait, actually, let's balance the equation with \( 4O_2 \). The left side: \( CH_4 + 4O_2 \), which has 1 C, 4 H, and 8 O. On the right side, for C: we need 1 \( CO_2 \) (1 C), for H: we need \( 2H_2O \) would give 4 H, but O from \( CO_2 \) is 2 and from \( 2H_2O \) is 2, total 4, but we need 8 O. So we need to double the \( H_2O \) to \( 4H_2O \) (which gives 8 O from \( 4H_2O \) (4×1 O) and 2 O from \( CO_2 \), total 10? Wait, no, let's do it properly. Wait, original with 2 \( O_2 \): left O is 4, right O is \( 2 + 2 = 4 \) (from \( CO_2 \) (2 O) and \( 2H_2O \) (2×1 O)). When we have 4 \( O_2 \), left O is 8. So right side: \( CO_2 \) has 2 O, so \( H_2O \) needs to have 6 O? No, wait, H: \( CH_4 \) has 4 H, so \( H_2O \) must have 4 H, so coefficient of \( H_2O \) is 2 (2×2 H), but then O from \( H_2O \) is 2, O from \( CO_2 \) is 2, total 4, but we need 8 O. So we need to double the \( H_2O \) to 4 (4×2 H, 4×1 O) and \( CO_2 \) to 2? Wait, no, let's balance the equation \( CH_4 + 4O_2
ightarrow?CO_2 +?H_2O \). C: 1, so \( CO_2 \) is 1. H: 4, so \( H_2O \) is 2 (2×2 H). O: left is \( 4×2 = 8 \), right from \( CO_2 \) is 2, from \( 2H_2O \) is 2, total 4. Not balanced. So we need to multiply \( H_2O \) by 4: \( 4H_2O \) gives 8 H (too much, since \( CH_4 \) has 4 H). Wait, I made a mistake. The number of H atoms is determined by \( CH_4 \), which is 4, so \( H_2O \) must have 4 H, so coefficient of \( H_2O \) is 2 (2×2 H). Then O from \( H_2O \) is 2, O from \( CO_2 \) is 2, total 4, but we have 8 O from \( 4O_2 \). So we need to have 2 \( CO_2 \) (2×2 O) and 4 \( H_2O \) (4×1 O), total O: 4 + 4 = 8. Wait, but then H: 4×2 = 8 H, but \( CH_4 \) has 4 H. Oh, right! I messed up. The number of H atoms is 4, so \( H_2O \) must have 4 H, so coefficient of \( H_2O \) is 2 (2×2 H). So O from \( H_2O \) is 2, O from \( CO_2 \) is 2, total 4. But we have 8 O from \( 4O_2 \). So this means that to balance O, we need to have 2 \( CO_2 \) (2×2 O) and 4 \( H_2O \) (4×1 O), but then H is 8, which is more than 4. So actually, the correct way is that if we increase \( O_2 \) to 4, we also need to increase \( CH_4 \)? No, the question is just changing the 2 in front of \( O_2 \) to 4, keeping \( CH_4 \) the same. Wait, that would make the equation unbalanced, but the question is about the change on the right side. Wait, maybe a simpler way: in the original equation, the ratio of \( O_2 \) to \( H_2O \) is 2:2 (or 1:1). So if \( O_2 \) is doubled (from 2 to 4), then \( H_2O \) should also be doubled (from 2 to 4) to maintain the ratio (since \( O_2 \) and \( H_2O \) are related in the reaction for H and O balance). Also, the \( CO_2 \) would remain 1? No, wait, no. Wait, the key is that in a chemical equation, the coefficients are in proportion. The original equation has \( 2O_2 \) and \( 2H_2O \). So if \( O_2 \) is multiplied by 2 (from 2…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
There are now 4 molecules of \( H_2O \) (the first option: "There are now 4 molecules of \( H_2O \)")